Achieve clarity on your biotherapeutic
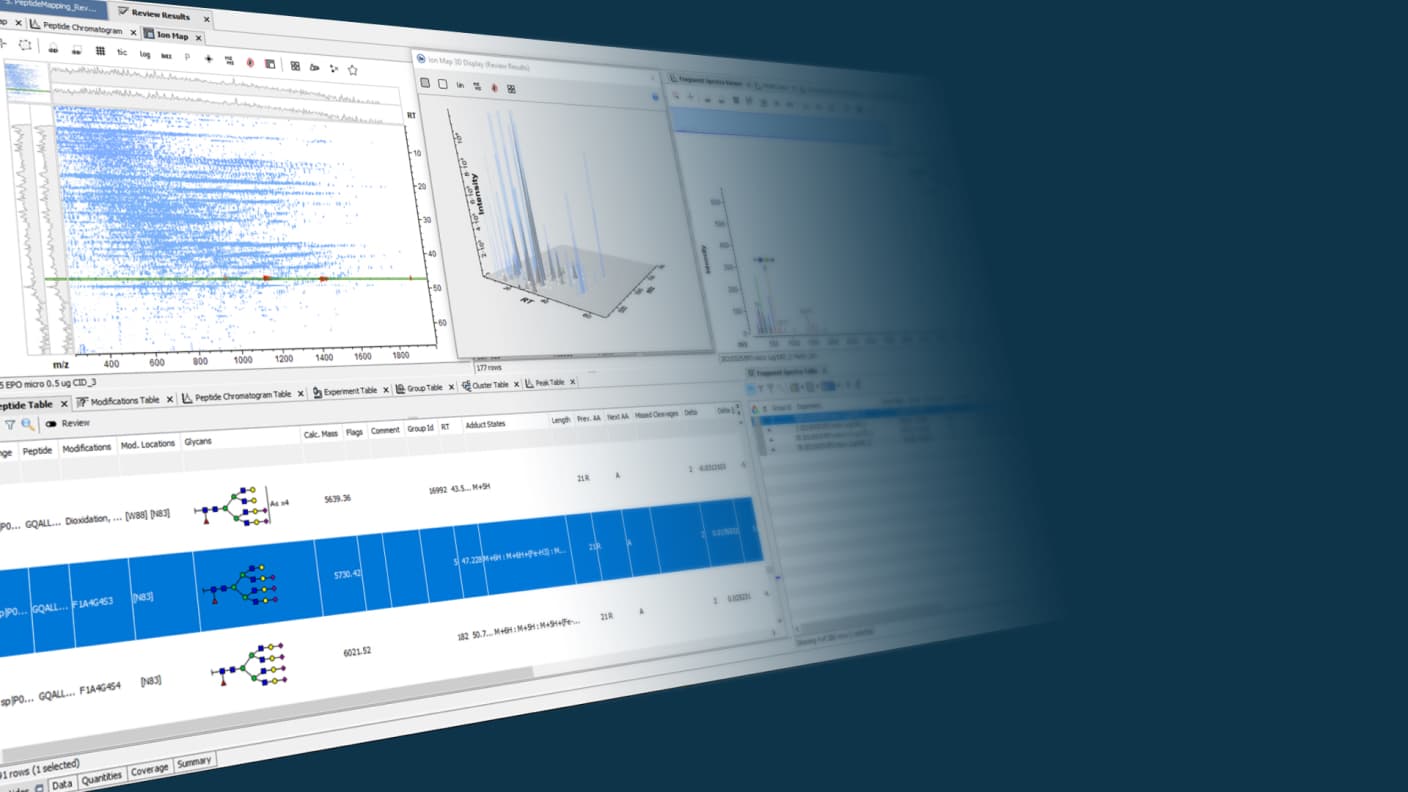
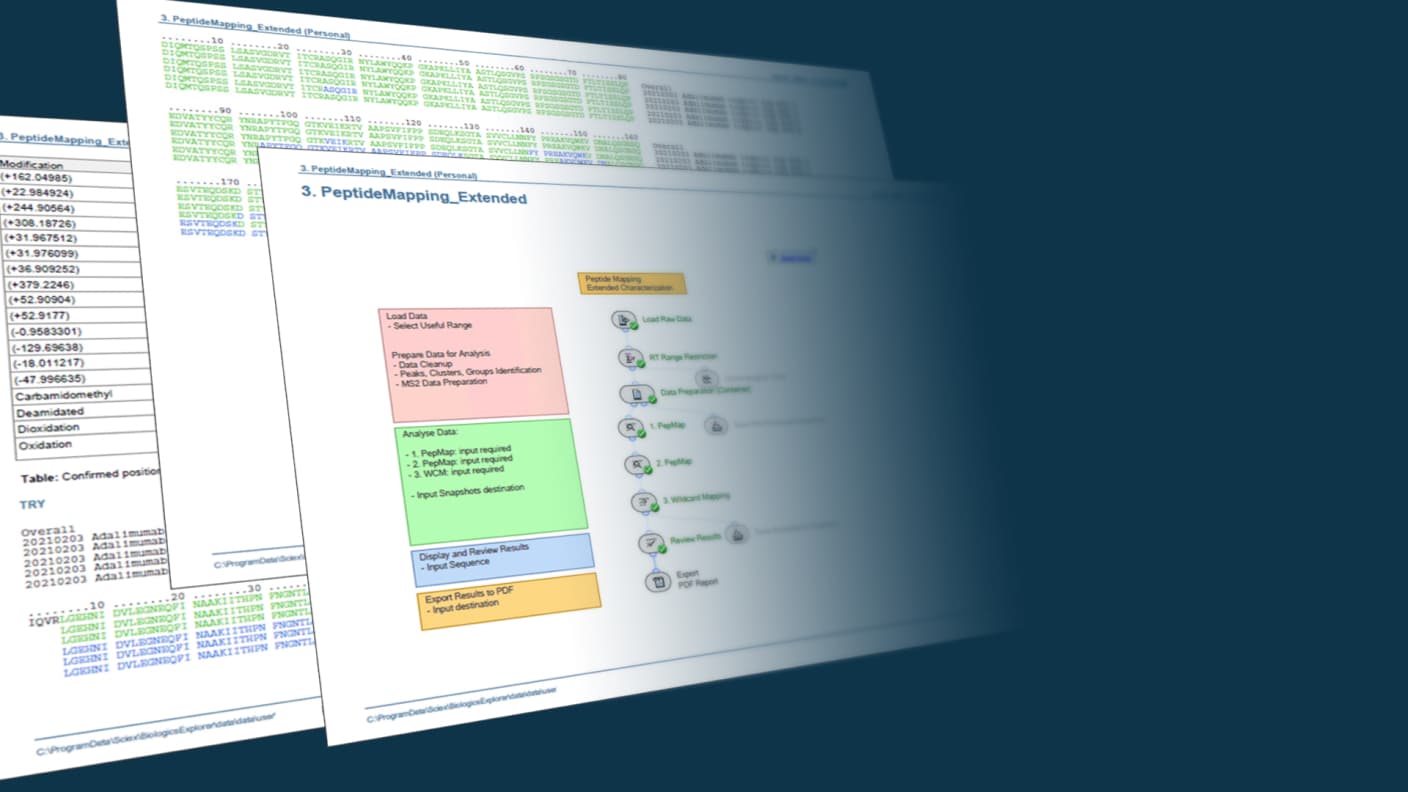
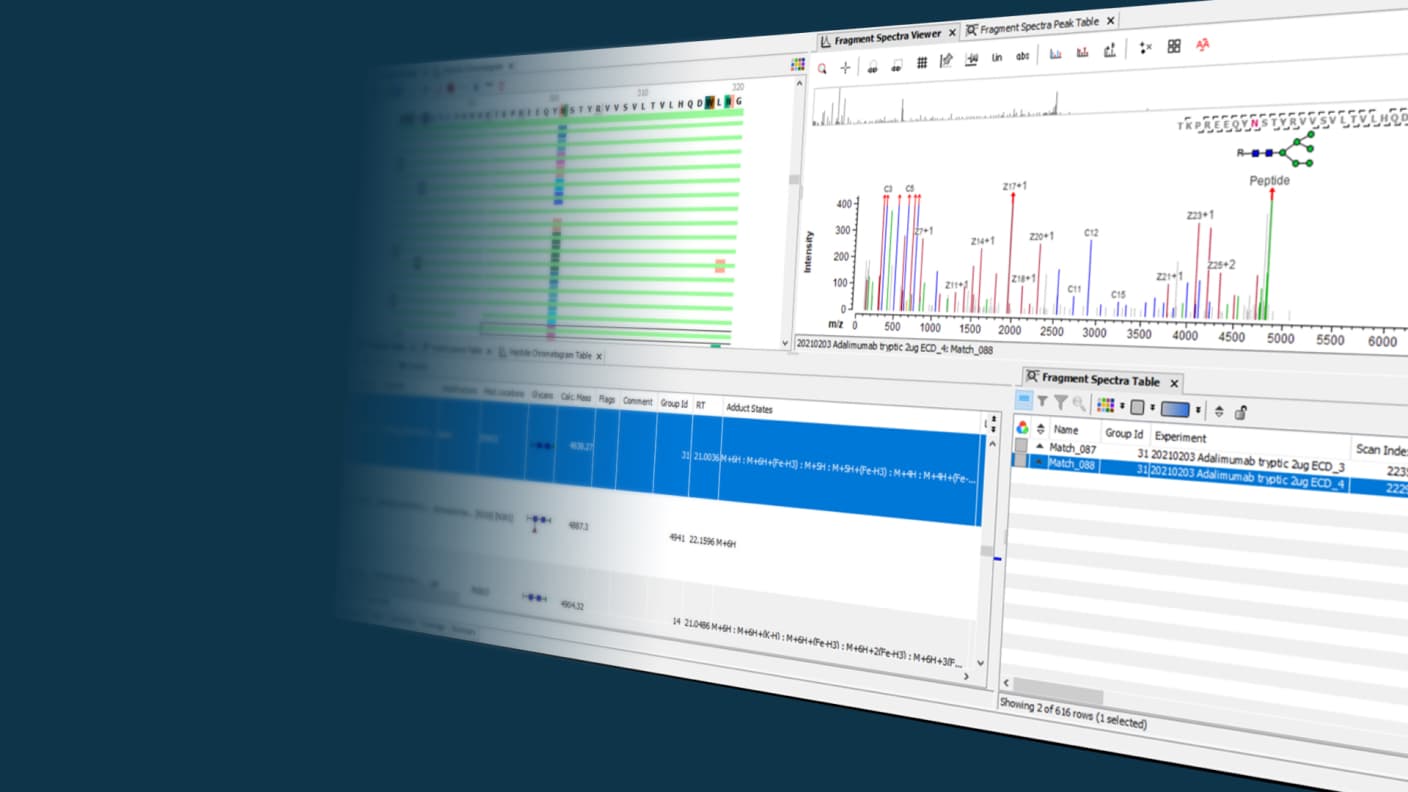
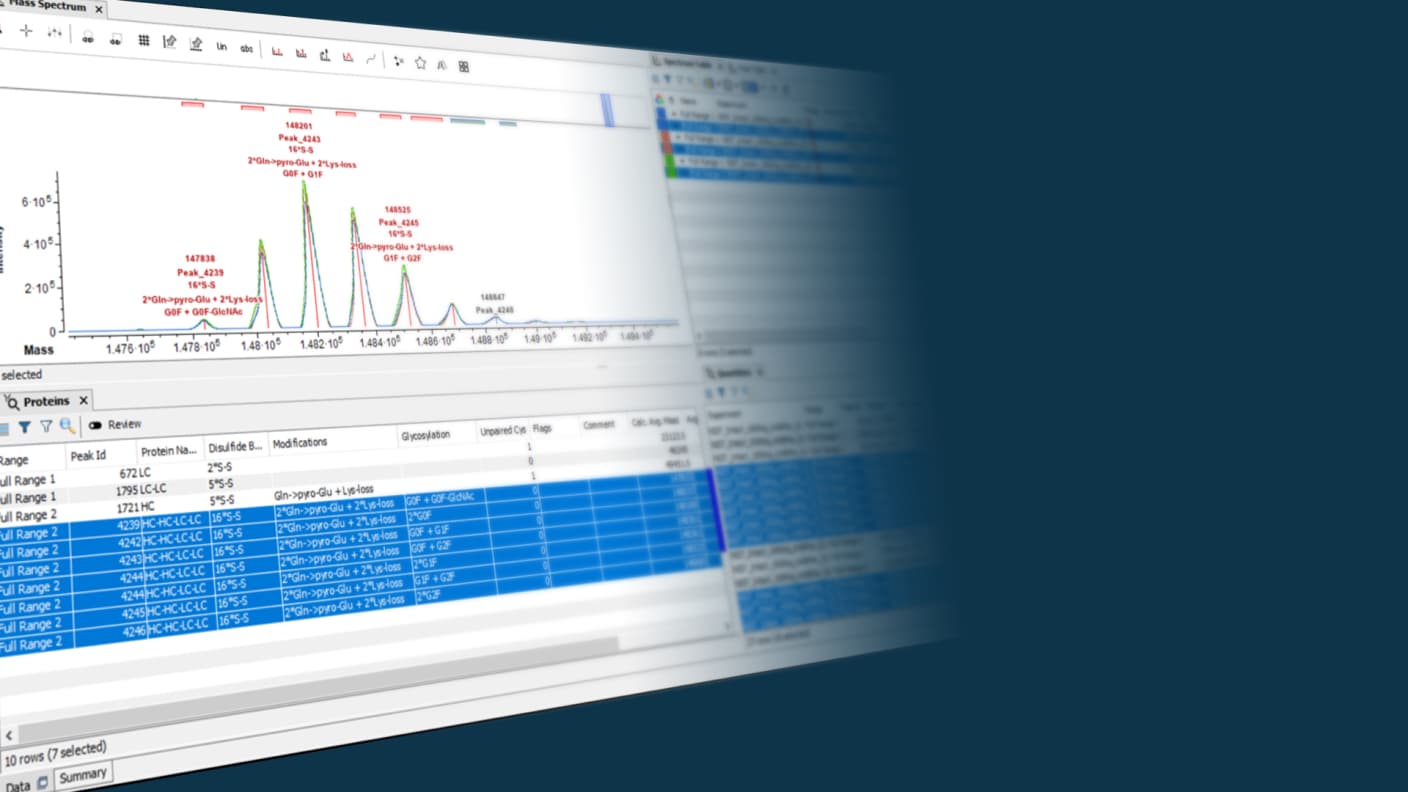
Leveraging the latest acquisition techniques and built on a heritage of proven performance, Biologics Explorer software delivers highly accurate and informative workflows for fully characterizing protein biotherapeutics. Reach decisions on the most important critical quality attributes for biopharmaceuticals faster and more confidently.
Biologics Explorer software is compatible with these featured systems below and across all SCIEX high resolution mass spectrometers:
A high-resolution mass spectrometry solution that combines powerful MS/MS sensitivity, fragmentation technology and a step-change in data independent acquisition.
A purpose-built QTOF system, designed specifically to accelerate everyday biologics characterization.
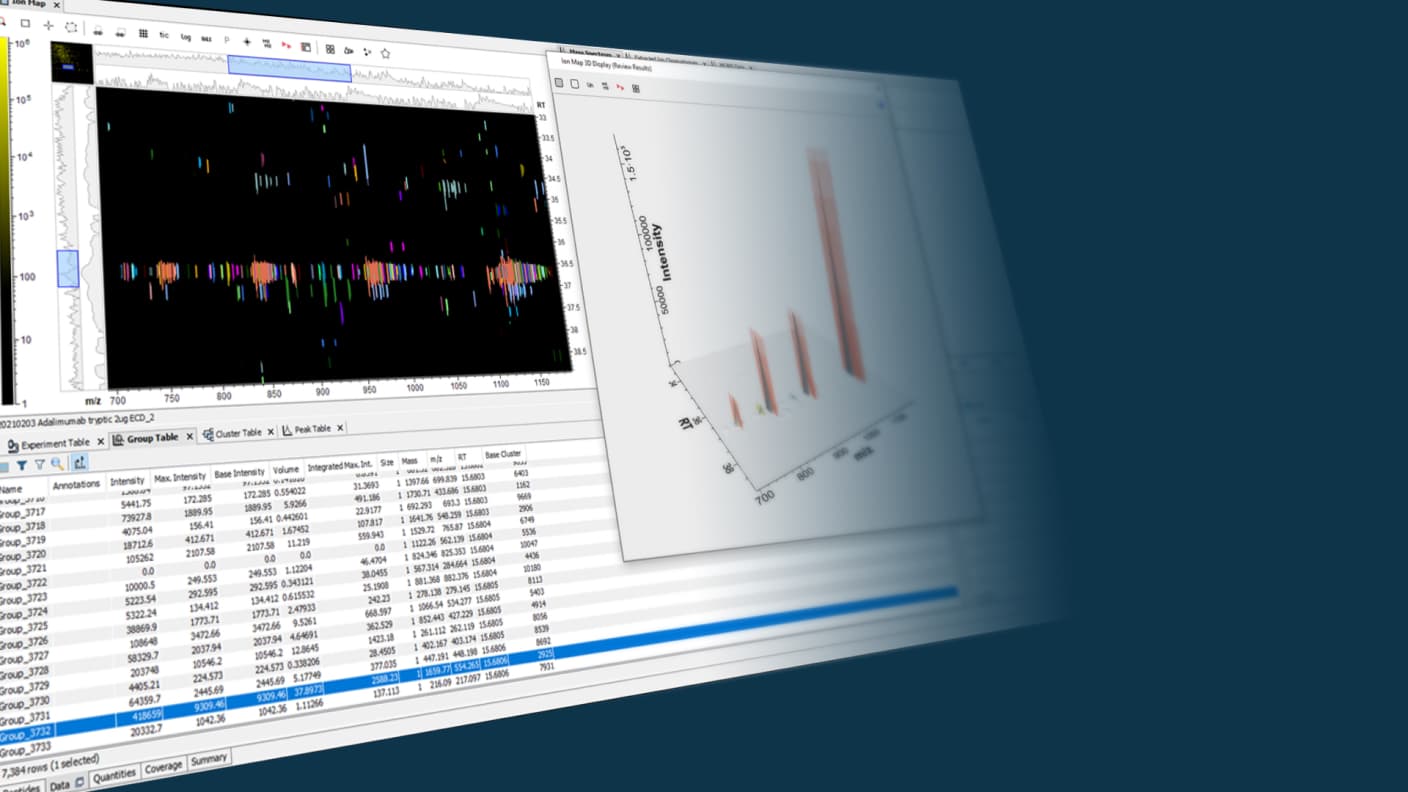
Eliminate the guess work and monitor biologics at the intact-protein level, gaining insights into charge variant-associated quality attributes.