Overview
Welcome to the Zeno revolution
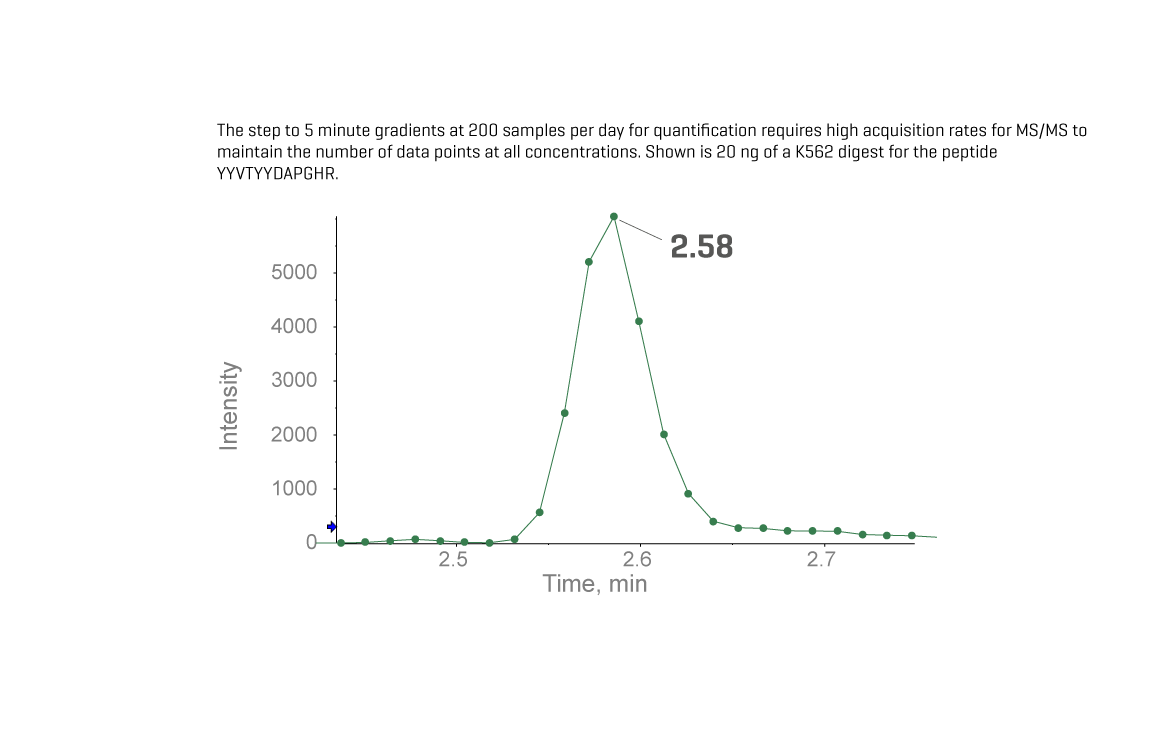
Driven by the power of the Zeno trap coupled with electron activated dissociation (EAD) fragmentation technology, this fragment-centric revolution unlocks sensitivity gains allowing you to uncover new information for certainty in your results to make better-informed decisions, faster.
Detect up to 20x more ions in every experiment and access a spectrum of tuneable fragmentation techniques to unlock new perspectives for every molecule, in every experiment.
Harness the power of Zeno SWATH data independent acquisition (DIA) to deliver a high depth of coverage, particularly on low abundance species, quickly and robustly. Routinely quantify up to twice the number of plasma proteins than previously possible and drive faster discovery of new disease markers and therapies.
“Zeno opens the door to a ‘fragment centric revolution’”
Maarten Dhaenens Ghent University, Belgium
The Zeno revolution is now...
Zeno trap, EAD and Zeno SWATH DIA. A high resolution mass spectrometry solution that combines MS/MS sensitivity, orthogonal fragmentation technology and a step change in SWATH DIA.
- Characterize large molecules including post-translational modifications
- Elucidate positional isomers on small molecules and lipids
- Identify and quantify proteins and peptides at unparalleled speed
- Gain significant sensitivity and discover less abundant ions
Overcome QTOF MS/MS duty cycle deficiencies
- >90% ions injected into the TOF
- Sensitivity gains of up to 5-20X with Zeno trap pulsing
- Identify and quantify low abundance species