Abstract
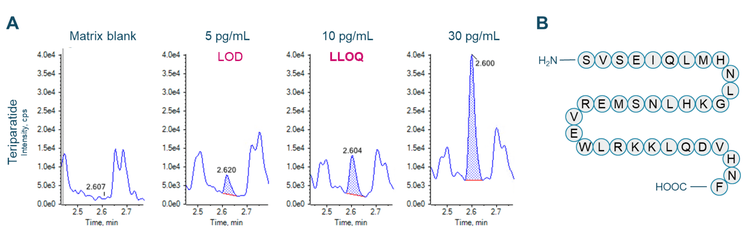
This technical note demonstrates a sensitive method to quantify teriparatide, a recombinant fragment of a human parathyroid hormone, on a SCIEX 7500 system. A lower limit of quantitation (LLOQ) of 10 pg/mL was determined in human plasma (Figure 1).
Introduction
Teriparatide comprises the first 34 amino acid residues of the human parathyroid hormone. Teriparatide has gained significant attention in osteoporosis management, given its ability to promote new bone formation and bone density enhancement at 20 μg/day. 1,2
Low-dose therapeutic drugs necessitate sensitive and selective assays for high-confidence detection and quantitation in biological matrices to ensure drug product safety and efficacy. Quantitation of peptides and proteins has commonly been performed using immunoassays such as ELISA. However, immunoassays inherently face analytical challenges, including poor selectivity, limited dynamic range and cross-reactivity. LCMS/MS methods are increasingly applied for therapeutic quantitation as they offer increased selectivity.
The presented method demonstrates a sensitive quantitation assay for teriparatide in human plasma. An LLOQ of 10 pg/mL level was achieved in human plasma using the SCIEX 7500 system. Front-end enhancements on the instrument facilitated improved overall ion generation, capture and transmission, providing optimal quantitative sensitivity.
Key features of the quantitation of teriparatide using the SCIEX 7500 system
- Low-pg/mL level quantitation of a recombinant parathyroid hormone fragment: Achieve a 10 pg/mL LLOQ for teriparatide in human plasma on the SCIEX 7500 system
- Ideal analytical performance: Achieve accurate quantitative performance with a high reproducibility of %CV <9% at all concentration levels
- Enhanced sensitivity unlocked: Improved front-end technology with the D Jet ion guide, OptiFlow Pro ion source and E Lens probe enhanced ion generation, capture and transmission, enabling users to reach desired quantitative sensitivity
- Streamlined data management: Data acquisition and processing are integrated into SCIEX OS software, a 21 CFR Part 11 compliance-ready platform
Methods
Sample preparation: The commercially available recombinant human parathyroid hormone fragment, teriparatide (Figure 1B) and 9-residue peptide analog leuprolide (Figure 2B) were reconstituted in dimethylsulfoxide. Teriparatide was spiked into 100 μL of human plasma at concentrations ranging from 10 pg/mL to 2000 pg/mL. Leuprolide was used as an internal standard (IS) and spiked at 20 pg. Protein precipitation was performed with 100 μL of 5% ammonia in 1:1 (v/v), water/ acetonitrile. Samples were vortexed for 30 seconds and centrifuged at 12000 rcf for 10 minutes at room temperature. The supernatant was transferred to a new Eppendorf tube containing 800 μL water and samples were briefly vortexed and centrifuged. A 500 μL aliquot of each sample was loaded twice on a Phenomenex Strata-X PRO micro elution solid-phase extraction plate operated under positive pressure. Samples were washed with 15% methanol in water. Finally, elution was performed using 1% trifluoroacetic acid in a solution containing 70% acetonitrile, 25% water and 5% trifluoroethanol by volume. The final elution volume was 100 μL.
Chromatography: Sample separation was performed using an ExionLC system at a 0.6 mL/min flow rate on a Phenomenex Kinetex XB C18 (2.1 x 50 mm, 1.7 µm, 100 Å) column. A 6- minute gradient was run using 0.1% formic acid in water as mobile phase A and 0.1% formic acid in acetonitrile as mobile phase B (Table 1). The column temperature was maintained at 55°C. An injection volume of 10 μL was used for analysis. A solution containing equal parts by volume of acetonitrile, methanol and water was used as a needle wash solvent.

Quantitative performance
This technical note demonstrates a low-pg/mL level quantitation of teriparatide in human plasma using the SCIEX 7500 system. Current methods require extensive sample preparation to meet sensitivity requirements and are often challenging with limited dynamic range. Here, the sample extraction method was optimized using a short and effective reverse-phase SPE procedure to minimize the matrix effects. In addition, chromatography and MS detection were optimized to develop a sensitive and selective bioassay in human plasma.
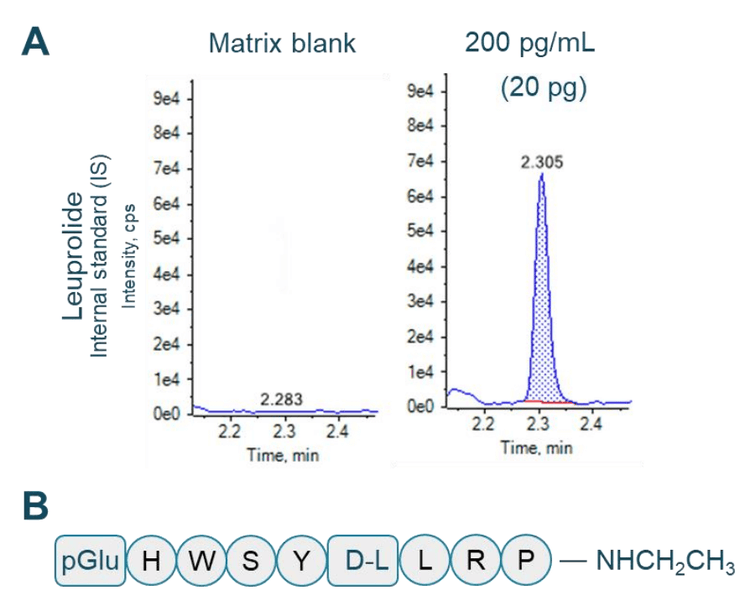
An LLOQ of 10 pg/mL was achieved for teriparatide (Figure 1). No interferences were observed at the retention times of teriparatide and leuprolide in human plasma (Figure 1A and Figure 2A).
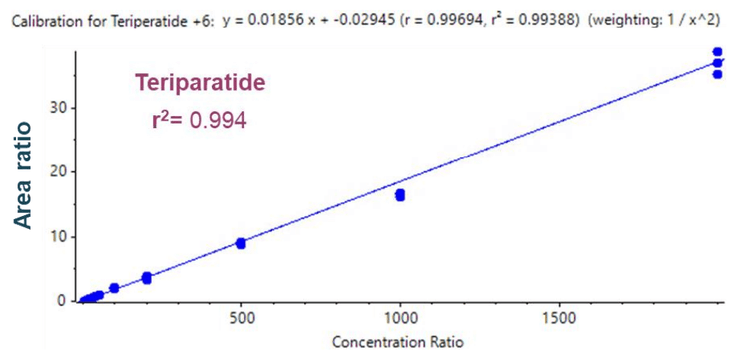
Linearity was achieved between 10 pg/mL and 2000 pg/mL with an r 2 of 0.994 (Figure 3).
Analytical performance was evaluated for accuracy and precision. The accuracy of the calculated mean was expected to be between 80% and 120% at the LLOQ and between 85% and 115% at higher concentrations. The %CV of the calculated mean for each concentration was expected to be <20% at the LLOQ and <15% at higher concentrations.
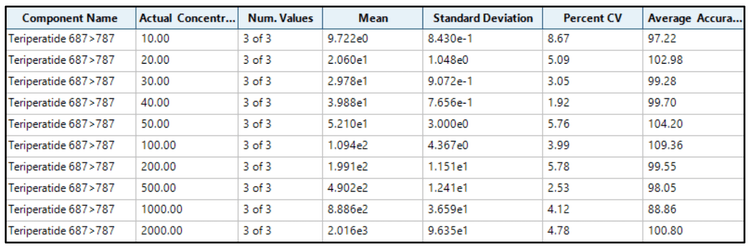
Accuracy was within ±10% of the nominal concentration and the %CV was <9% for teriparatide (Figure 4). Calculated accuracy and %CV values met the acceptance criteria at each concentration level.
Compliance-ready SCIEX OS software
SCIEX OS software is a closed system and requires records and signatures to be stored electronically, meeting the regulations outlined by 21 CFR Part 11. SCIEX OS software can open raw data files from any visible storage location within a closed network by using designated processing workstations. Figure 5 illustrates the features of SCIEX OS software used for monitoring the audit trail, acquiring and processing data and configuring user access.
The audit trail feature enables users to audit critical user actions and locks in data integrity. The Central Administrator Console (CAC) feature allows users to centralize acquisition and processing using a single platform to maximize efficiency for multi-instrument laboratories, independent of compliance standards. The configuration module allows users to assign roles and access as the administrator, method developer, analyst and reviewer.

Conclusion
- An LLOQ of 10 pg/mL was reached for the quantitation of teriparatide in human plasma
- Linearity was achieved between 10 pg/mL and 2000 pg/mL, with an r2 of 0.994
- The method demonstrated accurate and highly reproducible (%CV <9%) quantitative performance at all concentrations
- Sensitivity was achieved on the SCIEX 7500 system with an improved front-end technology for better ion generation, capture and transmission
- SCIEX OS software is compliance-ready to support 21 CFR Part 11 and integrates with a nominal mass spectrometer to support data acquisition, processing and management on a single platform
References
- Donald Bodenner, Carolyn Redman, Ann Riggs. Teriparatide in the management of osteoporosis. Clin Interv Aging. 2007;2(4):499-507.
- Julie Satterwhite, Michael Heathman, Paul D Miller, Fernando Marín, Emmett V Glass, Harald Dobnig. Pharmacokinetics of teriparatide (rhPTH[1-34]) and calcium pharmacodynamics in postmenopausal women with osteoporosis. Calcif Tissue Int. 2010 Dec;87(6):485-92.
- Richard G Kay, Jonathan T Hands, Glen Hawthorne, Scott Constable, Martin Grosvenor, Johannah A Sharman. Validation of an ultrasensitive LC-MS/MS method for PTH 1- 34 in porcine plasma to support a solid dose PK study. Bioanalysis. 2015;7(12):1435-45.

