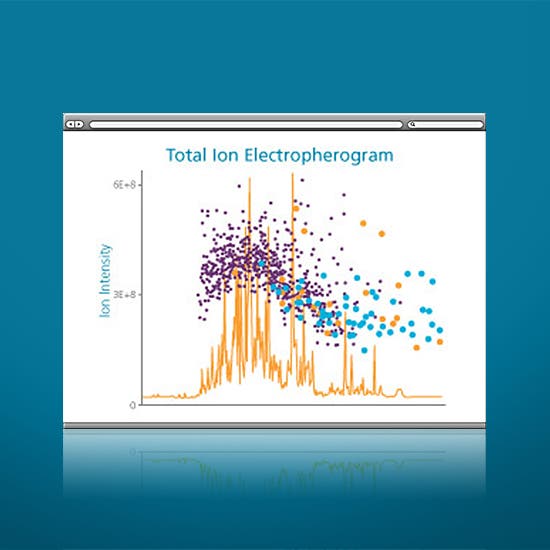
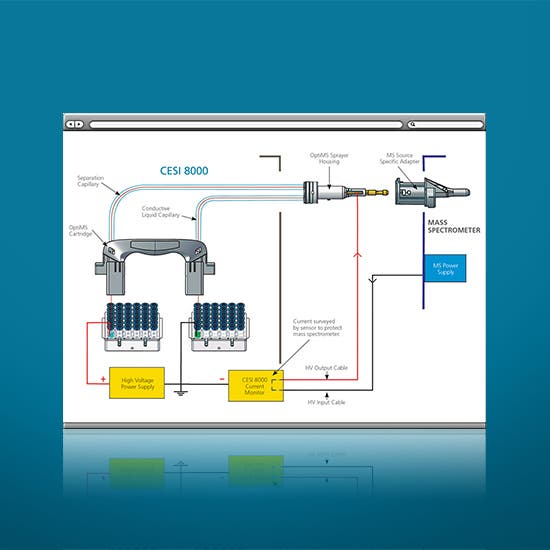
Experience the revolutionary ESI-MS approach to discovering previously undetectable characteristics from the minutest amount of precious samples, faster than ever before. Whether using as a standalone capillary electrophoresis instrument or in conjunction with the power of mass spectrometry, the CESI 8000 Plus system allows you to concentrate on your science rather than technology. You’ve worked hard to get your sample this far. Get the most from that precious material and hunt down the hidden information you couldn’t see before:
- Intact mAb charge variants with online MS detection
- Proteoforms and peptide PTMs such as those with multiple phosphorylation sites
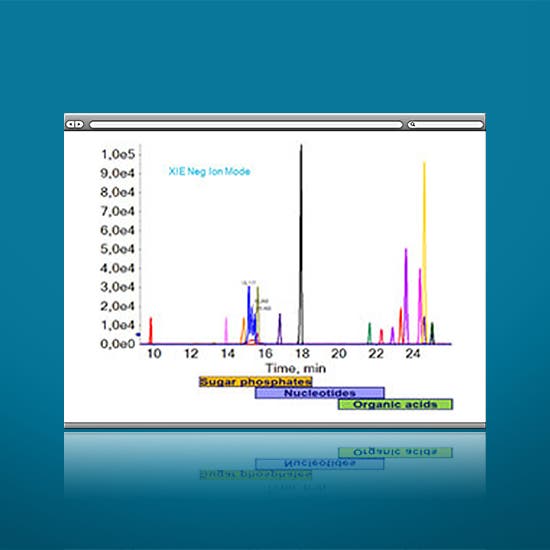
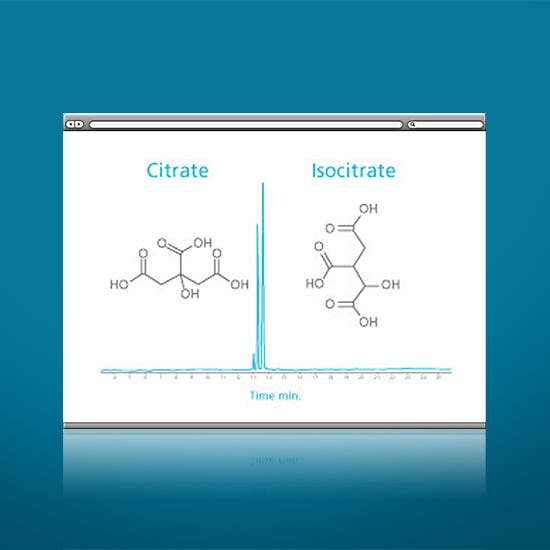
- Challenging charged and polar metabolites/degradation products such as anionic and/or hydrophilic analytes
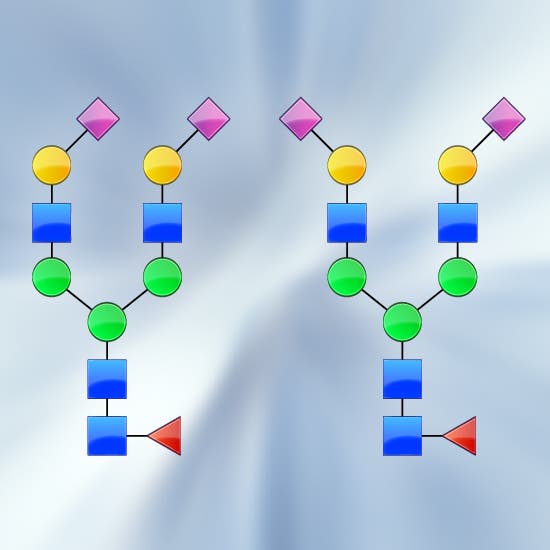
- Isobaric metabolites and glycans not resolved by traditional techniques
- Protein conformation and quantitation