Abstract
Sensitivity for two glycosphingolipids (GluSPH and GalSPH) were evaluated in cerebral spinal fluid using the SCIEX 7500 system. An assay was developed using HILIC chromatography and using positive MRM analysis and electrospray ionization. Calibration curves were generated on the SCIEX 7500 system as well as the QTRAP 6500+ system to assess the sensitivity difference.
Introduction
GalSPH and GluSPH are useful biomarkers for several diseases such as Krabbe and Gaucher disease as well as being a potential marker for carriers of heterozygous mutated GBA1 gene, which increases the risk of Parkinson’s.1,2 Therefore, it is important to be able to accurately monitor these compounds within the brain. Cerebral spinal fluid (CSF) is analyzed for GalSPH and GluSPH due to being the closest matrix available to monitor changes in the brain. However, the volume available is extremely limited as typically several biomarker assays need to be performed on a single sample. Therefore, it is important to have as much sensitivity as possible to ensure that lower volumes of CSF can be used.
Currently, this analysis is performed by Ardena Bioanalysis Assen using the SCIEX Triple Quad 6500+ system but, due to the increased sensitivity available with the SCIEX 7500 system, a comparison study to evaluate impact was performed. The objective was to assess whether the increase in sensitivity allows for lower CSF volumes to be analyzed.
The analysis was performed using HILIC chromatography to provide separation between the two isomers (Figure 1), with the triple quadrupole mass spectrometers being operated in positive mode (electrospray ionization) using MRM acquisition.
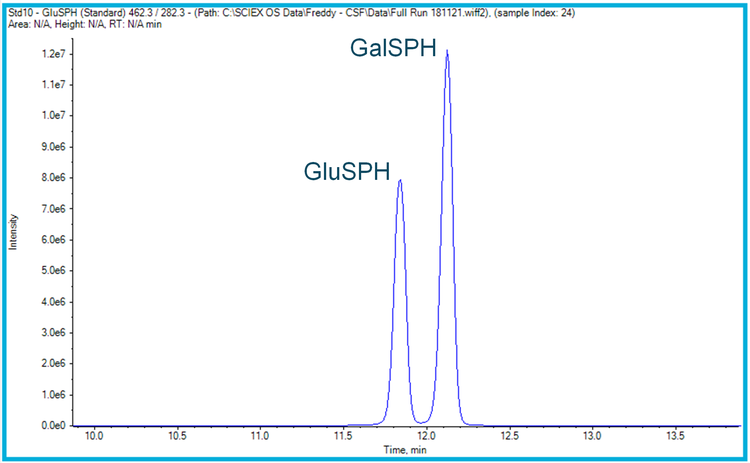
Methods
Sample preparation: Standards for galactosyl sphingosine (GalSPH) and glucosyl sphingosine (GluSPH) and their deuterated internal standards, GluSPH-D5 and GalSPH-D5, were obtained from Avanti Polar Lipids. Extracted standards and cerebral spinal fluid (CSF) samples were prepared by Ardena Bioanalysis. Standards were prepared in surrogate CSF across a range of concentrations suitable for analyzing endogenous levels of the CSF samples, with the deuterated internal standards held add a constant concentration. The samples were then split to be analyzed on both the SCIEX Triple Quad 6500+ and SCIEX 7500 systems.
LC-MS analysis: Samples were analyzed using both the QTRAP 6500+ system and the SCIEX 7500 system. Separation of Gal-and Glu-SPH was achieved using HILIC chromatography. MRM analysis was performed using electrospray ionization in positive polarity.
Data processing: Datasets were processed using both SCIEX OS software and Analyst software.
Results
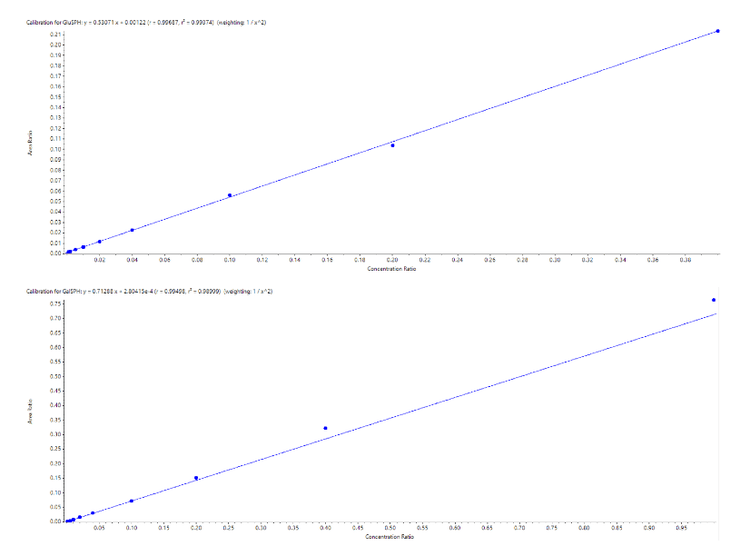
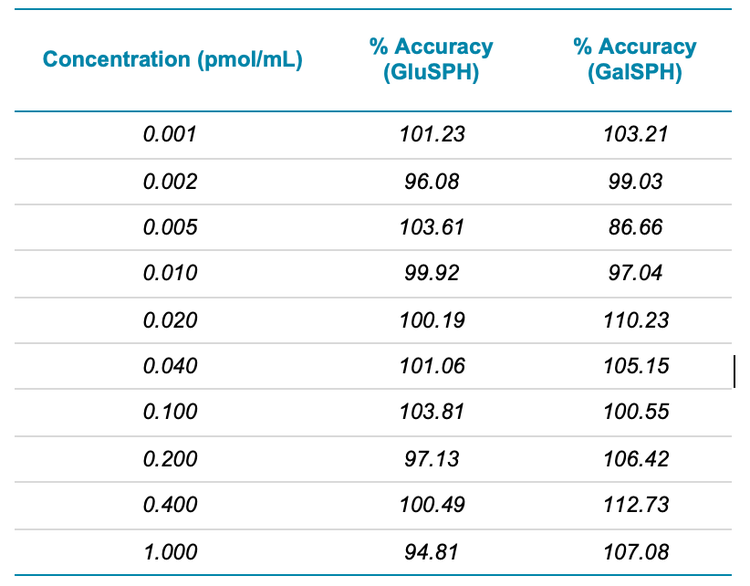
To compare the sensitivities between the two systems, a set of standards and CSF samples were prepared by Ardena Bioanalysis Assen before being divided and analyzed on both MS systems. Figure 2 highlights the calibration curves obtained for GluSPH and GalSPH on the SCIEX 7500 system across a concentration range between 0.001 – 1.000 pmol/mL. Both provided an r value >0.99, with accuracies between 80 – 120% at each level (Table 1), providing good linearity across the 3 orders of dynamic range studied. A similar concentration curve was generated on the QTRAP 6500+ system (data not shown).
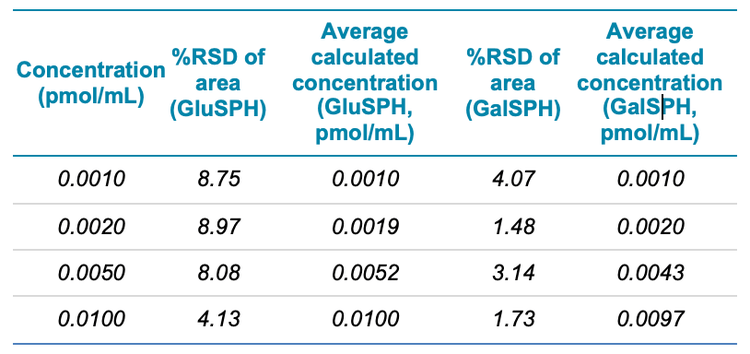
The %RSD and calculated concentrations for the bottom four surrogate CSF standards are also detailed in Table 2 highlighting that the %RSD values are below acceptable criteria of 10%.
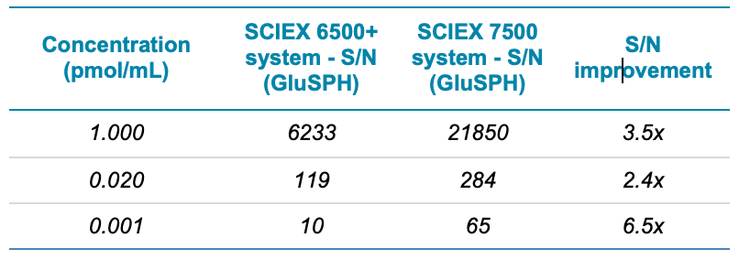
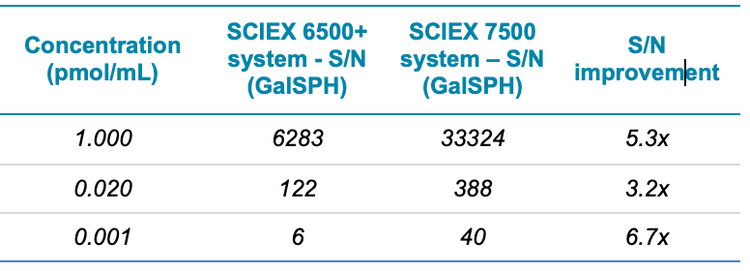
The sensitivity difference between the two systems was next assessed by using peak-to-peak S/N with similar noise regions, across the concentration curves. A summary is shown in Table 3 for GluSPH and Table 4 for GalSPH. The S/N increase was found to be between 2.4 and 6.7x, with improvements seen across the concentration range analyzed.
Conclusions
- Sensitivity for two glycosphingolipids, galactosylsphingosine (GalSPH) and glucosylsphingosine (GluSPH) were evaluated in cerebral spinal fluid (CSF)
- Two MS systems were evaluated, the QTRAP 6500+ system was compared to the SCIEX 7500 system
- Improvements in S/N of between 2.4 and 6.7 x were achieved on the SCIEX 7500 system, as compared to the QTRAP 6500+ system
- Good accuracy and linearity were observed across the range evaluated on the SCIEX 7500 system.
- Improved S/N at similar concentrations of the standards in matrix suggests that lower volumes of CSF could be used in this assay on the SCIEX 7500 system, this hypothesis will be evaluated next.
- Based on the results, it is concluded that the objective to increase the sensitivity is met. Based on the improved S/N seen on the SCIEX 7500 system when compared to similar analysis on the SCIEX Triple Quad 6500+ System it may be possible to reduce the required assay volume of CSF for this method when using the SCIEX 7500 system.
References
- Rohini Sidhu et al. (2018). A HILIC-MS/MS Method for Simultaneous Quantification of the Lysosomal Disease Markers Galactosylsphingosine and Glucosylsphingosine in Mouse Serum. Biomedical Chromatography. 32(7). e4235.
- Matthew Surface et al. (2021). Plasma Glucosylsphingosine in GBA1 Mutation Carriers with and without Parkinson's Disease. Mov Disord. 10. 1-6.
