A phenomenon called eutrophication promotes the natural occurrence of harmful cyanobacterial blooms in freshwater and marine ecosystems. (4) These blooms produce toxic metabolites that have diverse structural and physicochemical properties. Frequently detected cyanotoxins include microcystins, nodularins, cylindrospermopsin and neurotoxins, which increasingly threaten the ecosystem and human health. (5) While these blooms of toxic cyanobacteria species are present in various surface water sources, the primary route of exposure for most people is through drinking water, which can result in consumption advisories from regulatory authorities. (6-8)
For example, the World Health Organization (WHO) provisional guideline for microcystin-LR (MC-LR) is a limit of 1 μg/L. (9) The US Environmental Protection Agency (EPA) 10-day drinking water health advisory for MC-LR is a limit of 0.3 μg/L for infants and children up to 6 years old, and 1.6 μg/L for adults. (10) In Canada, the maximum acceptable concentration (MAC) of MC-LR is 1.5 μg/L. (11)
Traditionally, surface waters have been analyzed for these toxins using high-performance liquid chromatography (HPLC) with ultra-violet (UV) detection or other methodologies such as gas chromatography (GC). However, these approaches have limitations, especially when it comes to potential cross-contamination, which could lead to a high risk of false positives.
A shift to mass spectrometry for detecting biotoxins
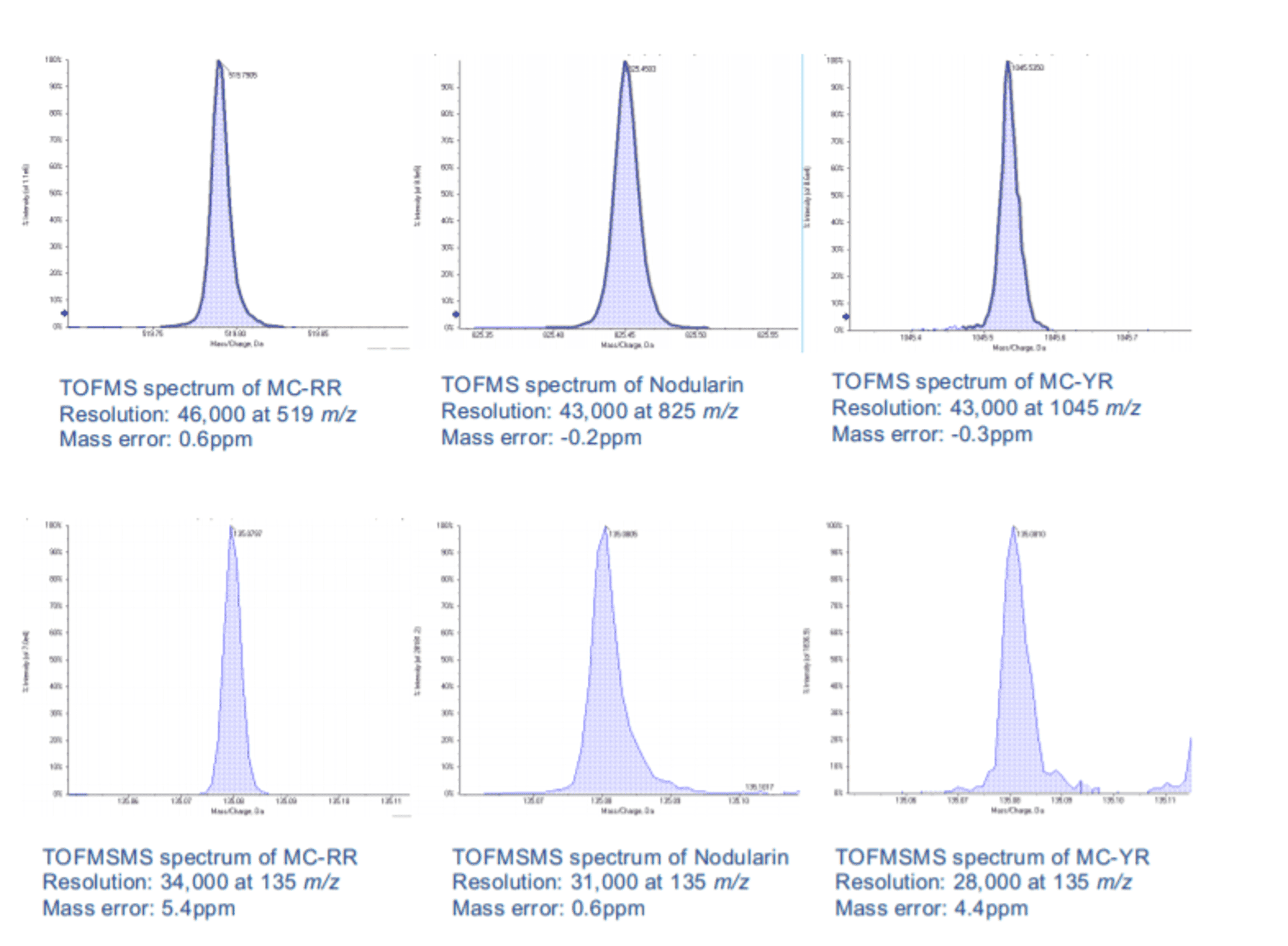
With the diversity in chemical structures of natural toxins in the environment, liquid chromatography-tandem mass spectrometry (LC-MS/MS) is an ideal tool for the comprehensive analysis of a wide scope of toxins, including microcystins, in a single analysis.
Using LC-MS/MS enables:
- Enhanced sensitivity for lower-level toxin detection
- Better performance by simplifying sample preparation and enabling a faster time to results
- Improved ruggedness to accommodate diverse sample types
- Higher specificity for the improved accuracy and reliability of results
To learn more about SCIEX solutions for mycotoxin and natural toxin detection in food and feed, click here.