Tunable electron activated dissociation (EAD) MS/MS to preserve particularly labile post-translational modifications
Site-localization of malonylated peptides using the SCIEX ZenoTOF 7600 system
Joanna Bons1, Jason Cason2, Birgit Schilling1, Christie Hunter3
1Buck Institute, USA, 2SCIEX, Canada, 3SCIEX, USA
Abstract
Determining the identity and the precise location of a post-translational modification on a protein is important to fully characterize function. Here, the utility of electron activated dissociation on the SCIEX ZenoTOF 7600 system was investigated for the characterization of labile post translational protein modifications. Leveraging the tunable kinetic energy, the malonyl modification on a synthetic peptide was preserved such that definitive localization was possible. In addition, the use of Zeno trapping provided significant sensitivity gains to ensure complete sequence characterization was obtained.

Introduction
Post-translational modifications (PTMs) are important players in a diverse group of functions that include protein conformation and signaling. Lysine acylation, such as malonylation, is one such PTM, and is regulated in part by lysine deacylases, which are members of the sirtuin (SIRT) protein family. In a previous study investigating SIRT5-regulated lysine malonylome, it was shown that 183 malonyllysine sites (from 120 proteins) out of the 1,137 identified sites (from 430 proteins) were significantly increased in Sirt5-/- KO versus wild-type mice.1 Specifically, it revealed that malonylation regulated GADPH activity. Malonylated peptides are however traditionally difficult to characterize using mass spectrometry and CID because the modification is extremely labile.
In this work, that involves using EAD technology with tunable kinetic energy, the effects of kinetic energy ramping on the preservation of highly labile PTMs (malonylation, for example) was studied, focusing on one previously identified malonyllysine site from GADPH (K-192). Two orthogonal fragmentation modes were compared (EAD vs. CID), to investigate the utility of each for PTM site localization. In addition, samples were measured using MRMHR mode to investigate the use of EAD for quantitative PTM characterization of labile modifications.2 MS/MS data were acquired with the Zeno trap activated, which provides significant sensitivity increases and enhances the quality of EAD MS/MS spectra.
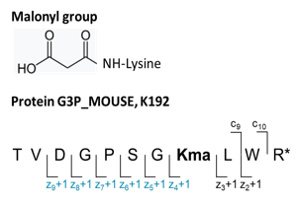
Figure 1. Sequence of the investigated malonylated peptide. Peptide at position 185-195 in mouse glyceraldehyde-3-phosphate dehydrogenase (P16858) and carrying a malonyl group on lysine K-192 was investigated. PTM site-specific ions are highlighted in blue.
Key features of SCIEX ZenoTOF 7600 system for PTM characterization
- ZenoTOF 7600 system enables acquisition of high-resolution MS and MS/MS spectra at high acquisition rates (up to 130Hz)
- Multiple fragmentation capabilities for flexibility: collision induced dissociation (CID) and electron activated dissociation (EAD)3
- Use of the Zeno trap for 5-10x increase in signal intensity on MS/MS fragment ions3
- Prevention of neutral loss from labile PTMs (no CO2-loss from malonyl modifications) using EAD MS/MS
- Tunable kinetic energy for EAD MS/MS using the EAD cell can be optimized and tailored for specific PTMs
- Efficient EAD product ion fragmentation generated strong ions for PTM site localization
- Preliminary assessment of EAD fragment ion quantification shows quantitative performance using high-resolution MRM (MRMHR) workflows
 Click to enlarge
Click to enlarge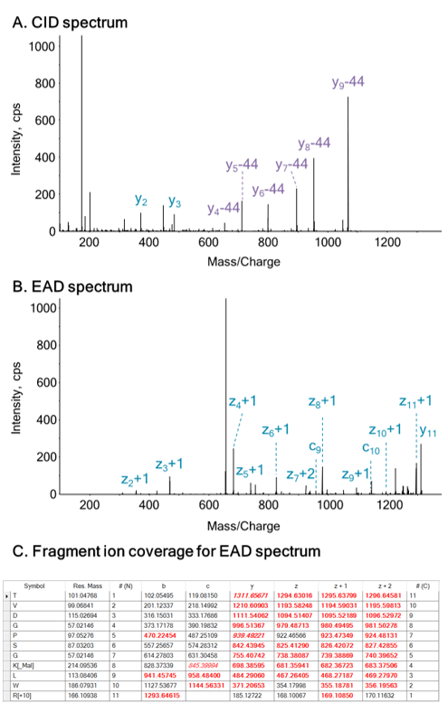 Click to enlarge
Click to enlarge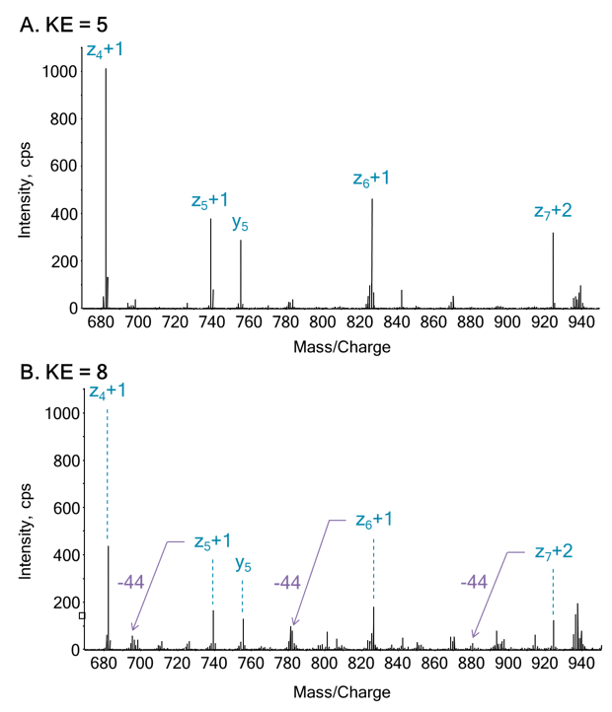 Click to enlarge
Click to enlarge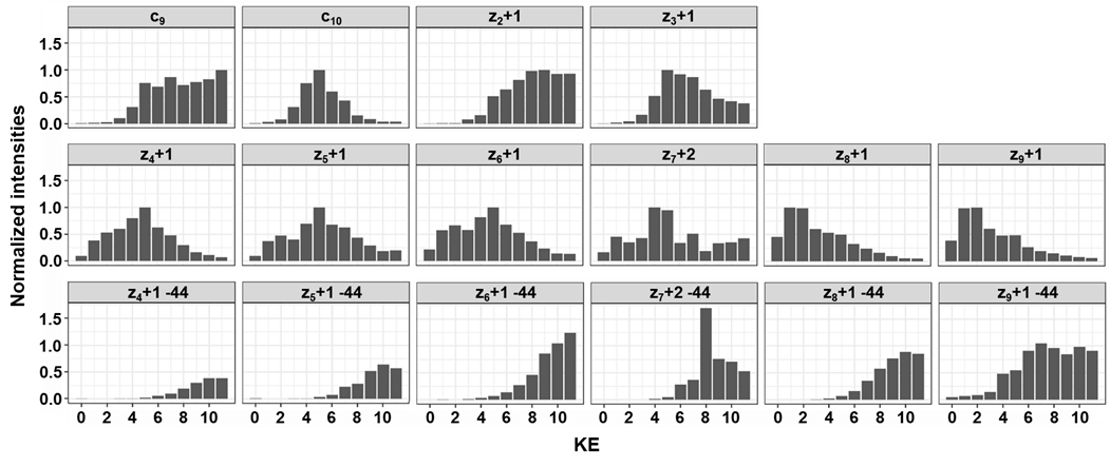 Click to enlarge
Click to enlarge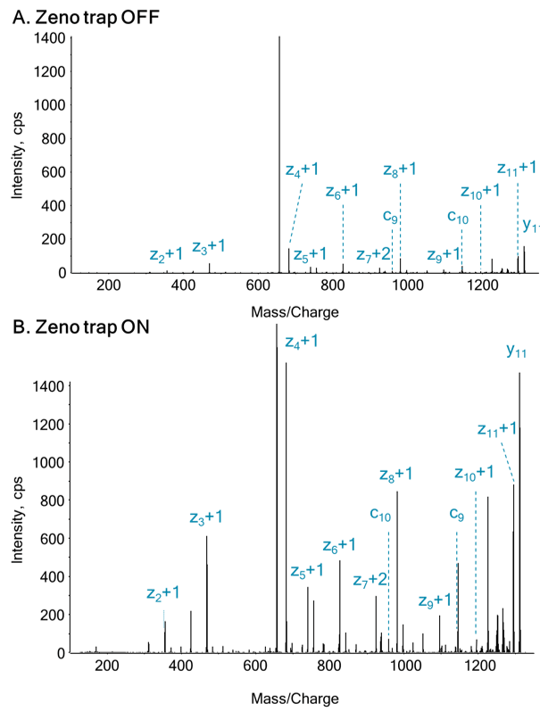 Click to enlarge
Click to enlarge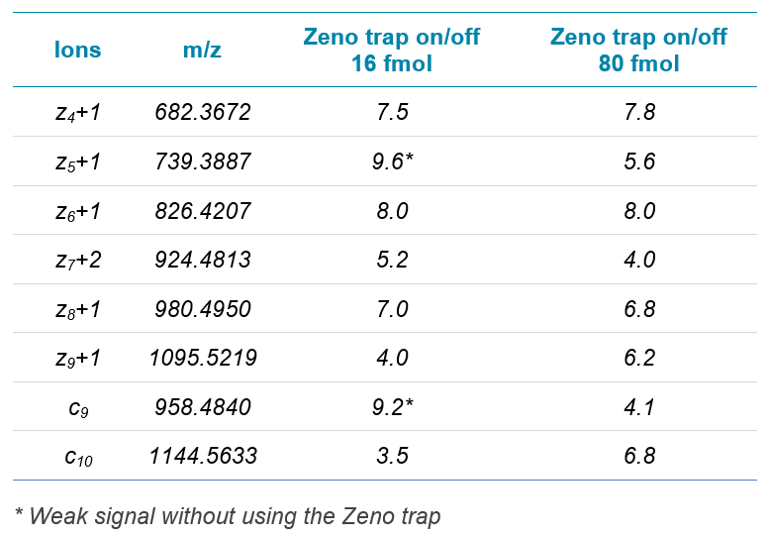 Click to enlarge
Click to enlarge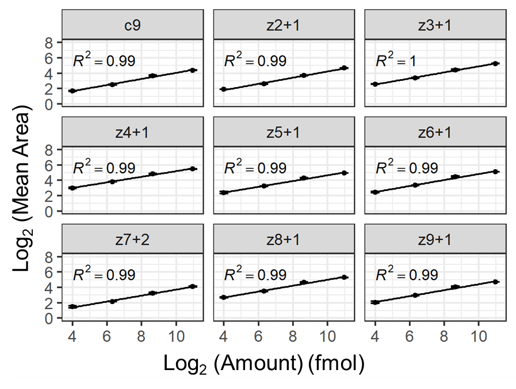 Click to enlarge
Click to enlarge