Confident quantification of per- and polyfluoroalkyl substances in water intended for human consumption
Using the QTRAP® 6500+ LC-MS/MS System and the SCIEX Triple Quad™ 7500 LC-MS/MS System – QTRAP® Ready
Kévin Privat,1 Carlos Bueno,2 Jianru Stahl-Zeng,2 Jack Steed,3 Philippe Diderich,4 Maria Isabel Beser5
1SCIEX, France; 2SCIEX, Germany; 3SCIEX, UK; 4AGE, Luxembourg; 5Salud Pública de Valencia, Spain
Abstract
An accurate and robust LC-MRM method using a smaller volume direct injection approach has been developed on the QTRAP 6500+ system, with sensitivity demonstrated to easily meet the current recommended limits set by the European authorities for drinking water (February 2020). Several PFAS specific analytical challenges have been addressed with this method, ensuring the method is easily implemented into labs. Data was also generated on the SCIEX 7500 system to highlight the advantages of increased sensitivity.

Introduction
Per- and polyfluoroalkyl substances (PFAS) are a family of thousands of synthetic compounds that are frequently found in the environment due to their widespread use in multiple industries. These compounds, which contain carbon-fluorine bonds, are incredibly resistant to breaking down. This means that they can accumulate within flora and fauna, and contaminate food and drinking water, leading to serious complications over time. Even if all manufacturing involving PFAS suddenly stopped, the existing environmental contamination would persist for decades to come. For this reason, it is paramount to limit the release of PFAS compounds into the environment. While some manufacturers have begun replacing PFAS compounds with either shorter-chain PFAS or non-fluorinated compounds, shorter-chain PFAS molecules have the potential for similar accumulation in the environment, food and drinking water.1
In February 2020, the European Parliament and Council of the European Union released a new directive that recasts the limit of PFAS in drinking water to 0.5 µg/L for all PFAS compounds identified, and 0.1 µg/L for a subset of PFAS compounds that are deemed particularly concerning for humans (see Table 1 for the LOQ of each compound analyzed). EU member states can choose which limit to use, and they can also decide to implement both. The difference between the limits is dependent on a list of compounds stated within the directive. The 0.1 µg/L limit applies to the compounds included in this list, which contain a perfluoroalkyl moiety with 3 or more carbons (i.e., –CnF2n–, n ≥ 3) or a perfluoroalkylether moiety with 2 or more carbons (i.e., –CnF2nOCmF2m−, n and m ≥ 1). The 0.5 µg/L limit applies to all PFAS compounds in totality.2
The requirements of this directive create the analytical challenge of achieving these newly set LOQs in drinking water without solid phase extraction (SPE) cleanup. This technical note demonstrates how these limits are easily achievable with a method that uses the QTRAP® 6500+ LC-MS/MS System for the analysis of all required PFAS compounds. In addition, it provides tips to help researchers achieve these limits and reduce common issues associated with the analysis without the use of specialized equipment or system modifications.
The conditions used with the QTRAP 6500+ System were also tested on the SCIEX Triple Quad™ 7500 LC-MS/MS System – QTRAP® Ready to highlight the increased levels of sensitivity that can be achieved with this system.
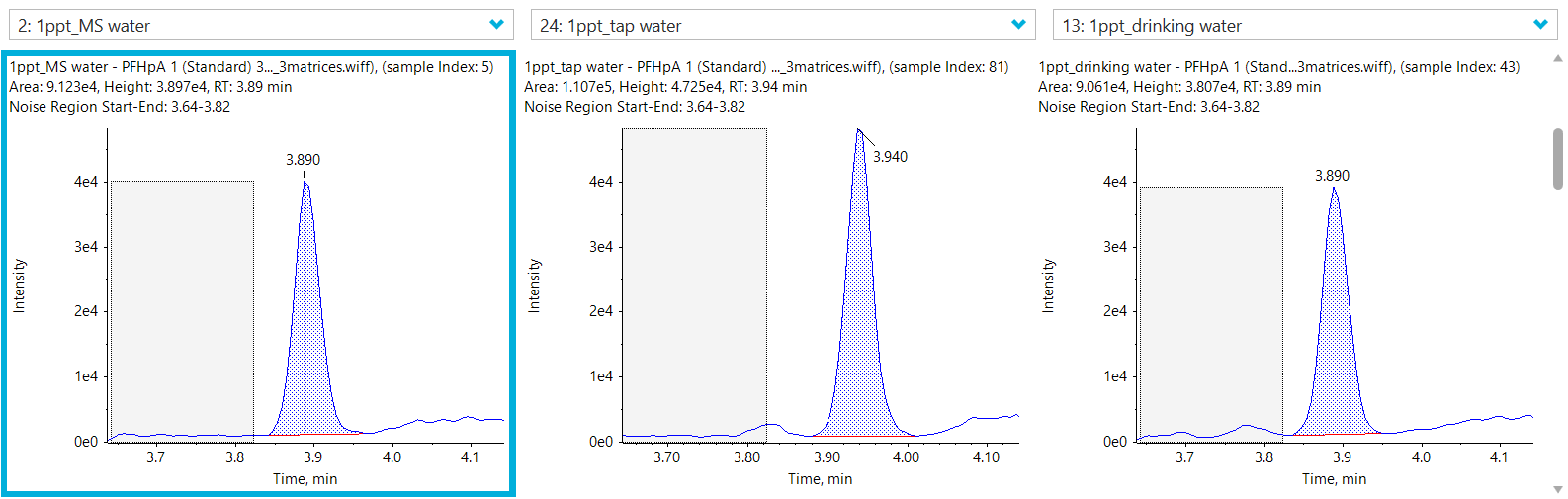
See Figure 1 for example XICs from a PFAS compound at it’s LLOQ in 3 different water matrices.
Figure 1. Extracted ion chromatograms (XICs) of PFHpA. XICs for PFHpA at the LOQ (0.001 µg/L) in 3 different water matrices: ultrapure HPLC-grade water (left), tap water (middle) and bottled mineral water (right).
Key features of the QTRAP 6500+ System for PFAS analysis
- LLOQ values down to 0.001 µg/L for the majority of the analyzed PFAS compounds
- The use of a 50 µL injection volume to meet the regulation requirement for drinking water (tap and bottled mineral water)
- Robust quantification with calibration curves for the vast majority of PFAS compounds between 0.001 and 1 µg/L
- No carryover or contamination in blank injections for analyzed PFAS compounds
- The use of a delay column to ensure that instrument contaminants are moved away from the analyte’s retention time so that accurate quantification can be achieved
 Click to enlarge
Click to enlarge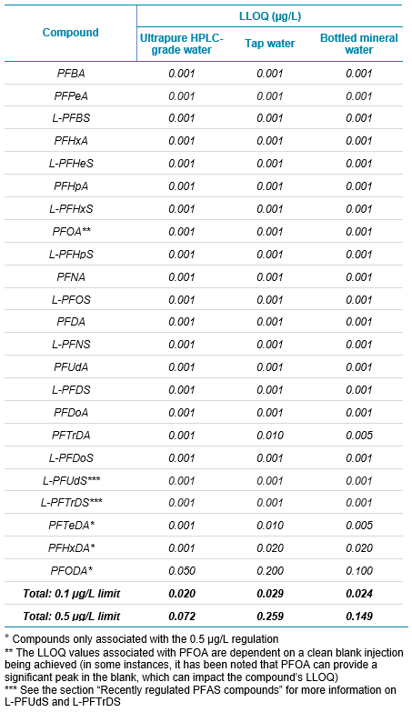 Click to enlarge
Click to enlarge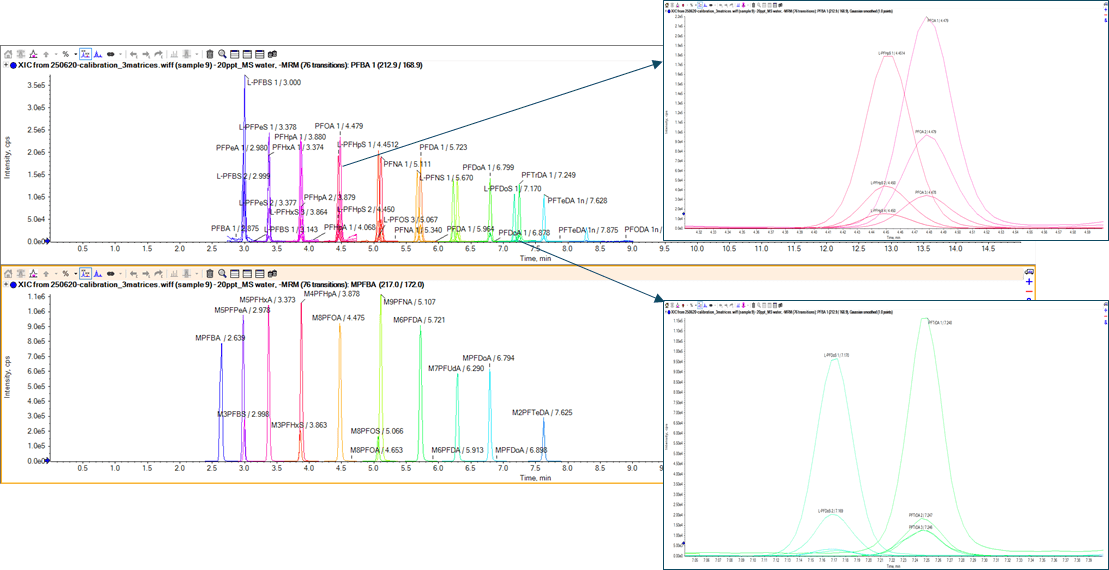 Click to enlarge
Click to enlarge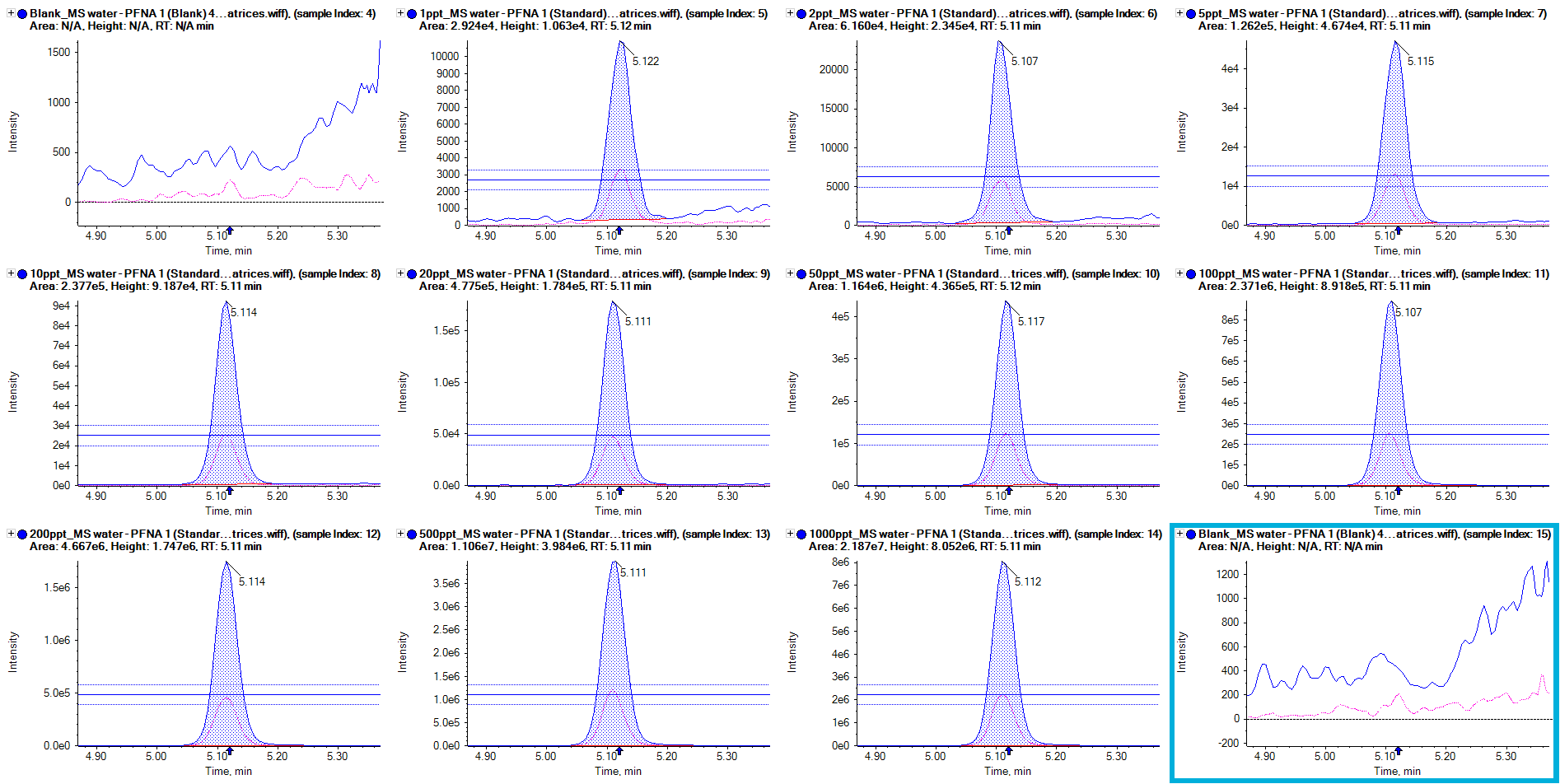 Click to enlarge
Click to enlarge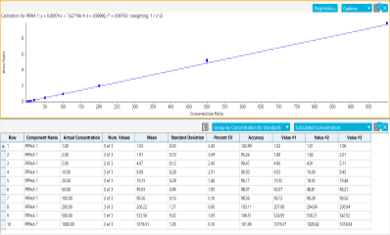 Click to enlarge
Click to enlarge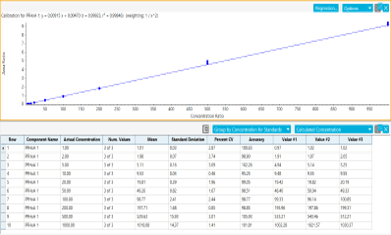 Click to enlarge
Click to enlarge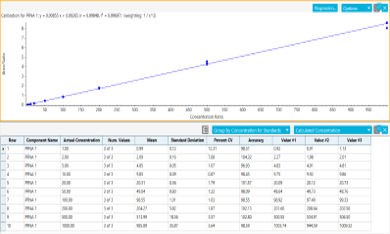 Click to enlarge
Click to enlarge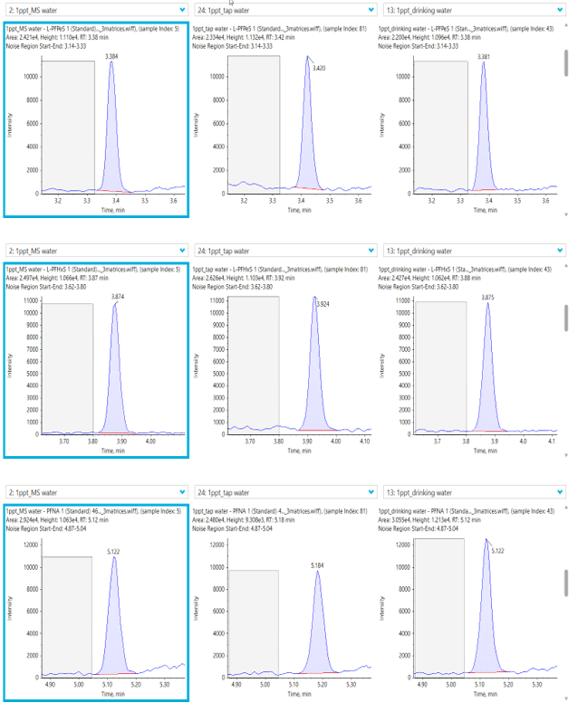 Click to enlarge
Click to enlarge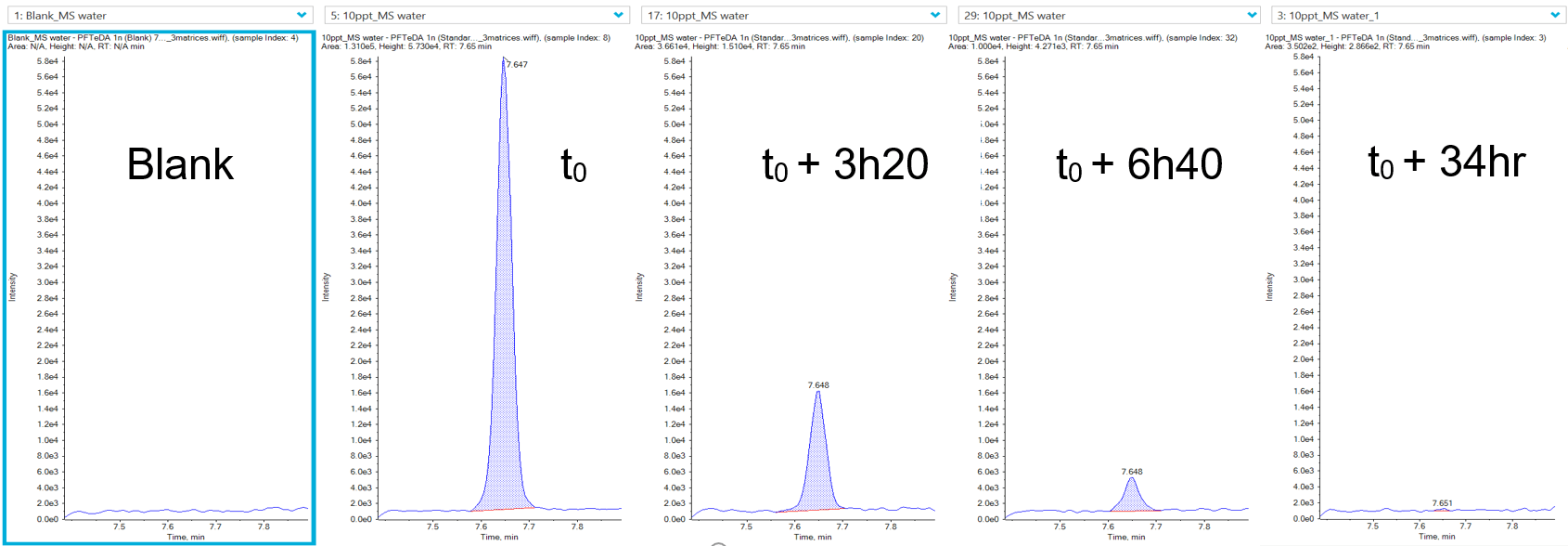 Click to enlarge
Click to enlarge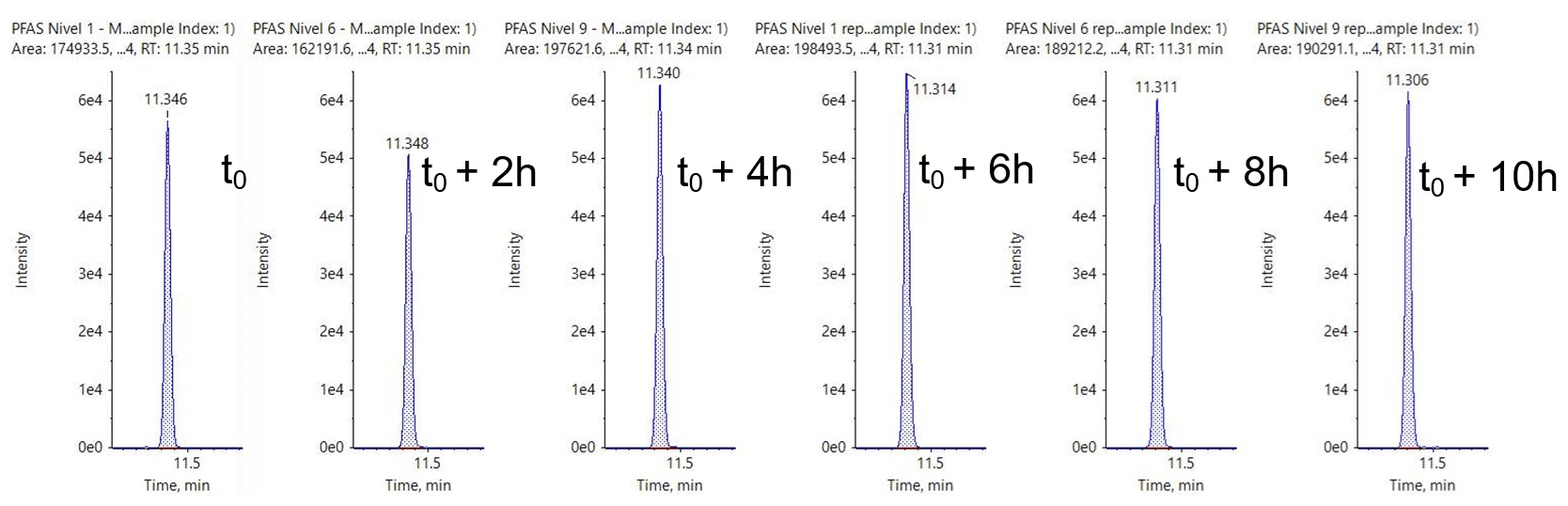 Click to enlarge
Click to enlarge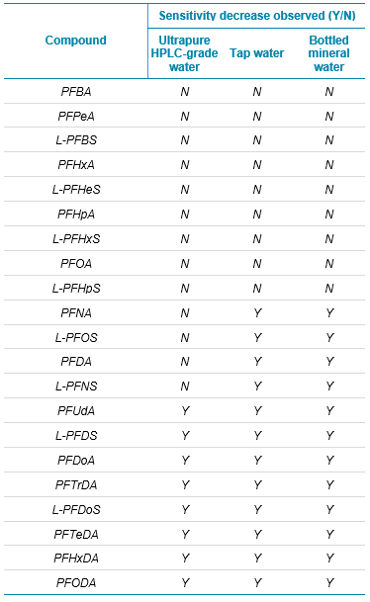 Click to enlarge
Click to enlarge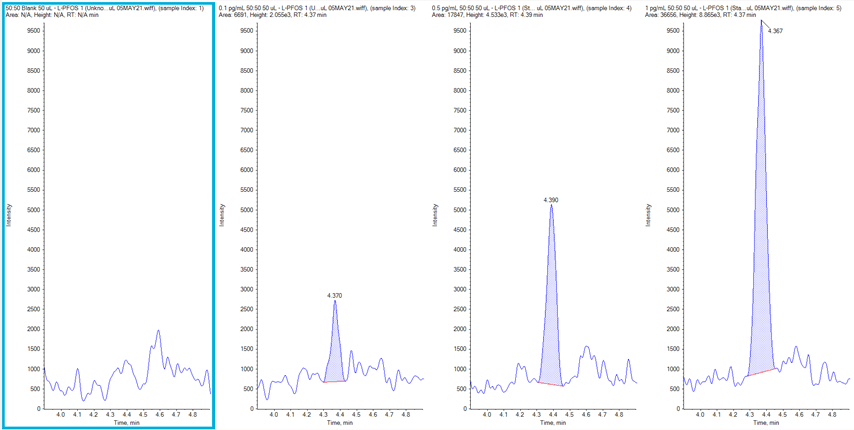 Click to enlarge
Click to enlarge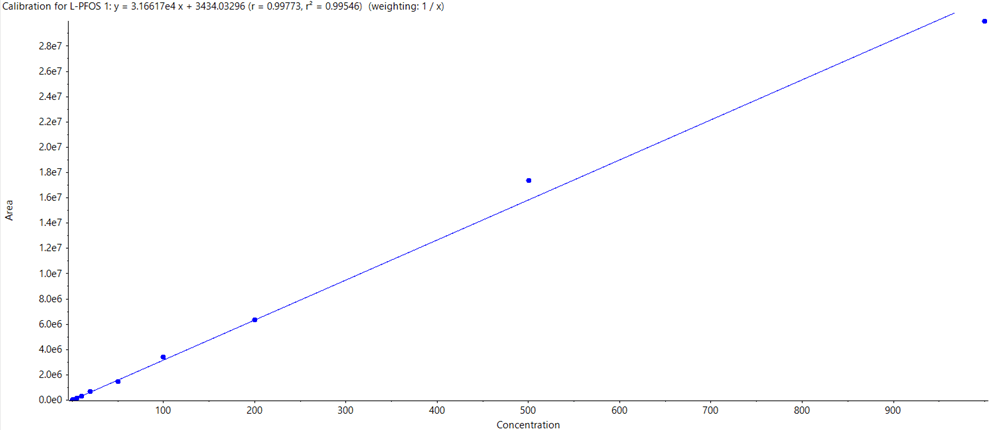 Click to enlarge
Click to enlarge Click to enlarge
Click to enlarge