Profiling and identification of hop-derived bitter compounds in beer using LC-MS/MS
André Schreiber
SCIEX, Canada
Abstract
X500R QTOF System was used to analyze 40 different beer, profiling the top 26 known hop-derived bitter compounds. Samples were processed using PCA in MarkerView™ Software to group and find discriminating features, highlighting the power of LC-MS/MS to study flavor and aroma profile in food and beverage samples,

Introduction
Beer is one of the world's most widely consumed beverage (after tea and water) and probably the oldest alcoholic beverage.1 Beer has attracted consumers over centuries due to its refreshing character, attractive aroma, and typical bitter taste.
The production of beer is called brewing, which involves the fermentation of starches, mainly derived from cereal grains (most commonly malted barley, although wheat, corn, and rice are widely used). Most beer is flavored with hops, which add bitterness and act as a natural preservative, though other flavorings such as herbs or fruit may occasionally be included.2 Aroma-active volatiles as well as nonvolatile bitter compounds of beers have been thoroughly investigated in recent decades.
The typical hop-derived beer bitterness is caused by adding cones, pellets, or extracts of hop during wort boiling. A few isomerization processes have been identified to be of major importance for bitter taste development in the final beer product. The so-called isoxanthohumol (Figure 1), identified as a bitter compound in beer, was found to be generated from the hop-derived xanthohumol, during wort boiling. Moreover, trans- and cis-iso-α-acids have been identified as the major bitter contributors in beer and were demonstrated to be generated upon a rearrangement reaction of their hop-derived precursors, the α-acids. Following the α-acids, the second major constituents of hop are the β-acids (Figure 1), but there are almost no data available on the direct contribution of these compounds to beer bitterness or on their role in the generation of bitter-tasting conversion products during wort boiling.3, 4
Figure 1. Hop-derived bitter compounds and their isomerization processes during the wort boiling.
General method for beer profiling
Here an LC-MS/MS based method is presented to identify hop-derived bitter compounds in beer.
- High resolution-accurate mass MS and MS/MS data were collected using the SCIEX X500R QTOF System.
- MS and MS/MS information were collected in a single injection using information dependent acquisition (IDA) for confident compound identification.
- Targeted and non-targeted data processing are demonstrated in workflows within the SCIEX OS Software and MarkerView™ Software.
- Principle component analysis in MarkerView Software allows for classification of sample groups and profiling based on target or unknown sample constituents.
- Compound Identification tools within the SCIEX OS Software include FormulaFinder algorithm and ChemSpider database. These can be used to tentatively identify candidate structures for non-targeted features.
Methods
Sample preparation: Beer samples were purchased from the Liquor Control Board of Ontario (LCBO). Samples were degasses and then diluted 2x with water. 5 µL of sample was injected.
Chromatography: Separation was performed using an ExionLC™ AD System, with a Phenomenex Luna Omega 1.6 µm Polar C18 (50 x 2.1 mm). A simple gradient was used as described in Table 1, at a flow of 0.5 mL/mim.
Table 1. Gradient conditions.
Mass spectrometry: The SCIEX X500R QTOF System with Turbo V™ Source and Electrospray Ionization (ESI) was used in positive polarity. Ion source temperature was set to 450ºC and IS voltage was set to 5500 V. Mass calibration was achieved using the integrated calibrant delivery system (CDS) with the TwinSprayer probe (dual ESI needle).
High resolution data were acquired using an information dependent acquisition (IDA) method consisting of a TOF-MS survey (100-1000 Da for 200 msec) and up to 10 dependent MS/MS scans (50-1000 Da for 50 msec). Declustering Potential (DP) was set to 80 V and MS/MS fragmentation was achieved using a Collision Energy (CE) of 35 V with a Collision Energy Spread (CES) of ±15 V.
Dynamic background subtraction (DBS) was activated to achieve the most complete MS/MS coverage. No inclusion list was used which allowed non-target identification without the need for a second injection to acquire MS/MS data. All data were acquired and processed using SCIEX OS Software.
Data processing: MarkerView Software 1.3 was used for statistical processing using Principal Components Analysis (PCA).
Figure 2. Example resolution data for isoxanthohumol. Resolution of 30000 or greater in both MS and MS/MS modes is achieved due to the N-optics design of the X500R QTOF System.
X500R System performance characteristics and data acquisition workflows
The X500R QTOF System utilizes N-optics design to maximize resolution while maintaining benchtop design and a minimized footprint. The resolving power increases with mass range providing ~30000 to 40000 resolution for the typical molecular weight range of ingredients and potential contaminants in beer (Figure 2).
The X500R QTOF System achieves stable mass accuracy of less than 2 ppm by using a heated TOF configuration, with 6 heater drones throughout the TOF path and by using the dynamic background calibration software algorithm. In addition, both dynamic transmission control and dynamic background calibration are used for further provide long term mass accuracy stability.
In addition, the integrated CDS with the TwinSprayer probe provides an independent calibrant delivery path for reliable auto-calibration (Figure 3), maintaining mass accuracy over long periods of time by automatically calibrating in batch mode.
Figure 3. TwinSprayer ESI probe. The unique design provides an independent flow path for calibration solutions, for easy maintenance of mass accuracy during LC-MS runs
Figure 4. Simultaneous acquisition of TOF-MS and MS/MS spectra using IDA. MS data alone is rarely sufficient for a definitive identification, therefore the ability to collect MS/MS data for ID in the same injection is desired. The example shows high quality spectra of isoxanthohumol (355) and the pesticide boscalid (343) collected during IDA.
The accurate measurement of a molecular ion is insufficient for compound identification. Single stage MS information can only be used for empirical formula finding. Accurate mass MS/MS data is necessary to identify chemical structures based on the pattern of fragment ions. Using IDA combined with the high acquisition rates of the X500R System, simultaneous acquisition of TOF MS and MS/MS data in a single data file for each sample was possible. Up to 10 MS/MS spectra were automatically collected for each chromatographic data point (Figure 4).
Processing workflow for target identification in SCIEX OS Software
A targeted processing method was built to profile 26 known hop-derived bitter compounds (Figure 5). Retention times were established by comparing the chromatographic profiles of beer samples to results published in literature.3
During targeted processing, extracted ion chromatograms (XIC) of all analytes are generated based on user input (chemical formula and expected retention time). MS and MS/MS information is automatically evaluated if an XIC signal is detected and compounds are identified by matching retention time, accurate mass and isotope pattern of the molecular ion.
During this project a small high resolution MS/MS library was generated to assist future identification by also utilizing the MS/MS fragmentation (Figure 6).
Figure 5. Targeted processing method to profile known hop-derived bitter compounds. XICs for each compound will be automatically generated based on provided chemical formula and retention time.
Figure 6. High resolution library for compound detection. For this work, a high resolution library for the detected bitter compounds was generated and can easily be added to SCIEX OS Software to assist similar studies.
Results of analyzing 40 beer samples
The amounts of xanthohumol (X) and isoxanthohumol (IX) in a beer can provide information about how hops were used during the beer making process. Figure 7 shows the intensity for IX and X in 3 different beers. The observed X/IX ratio of the pilsner and Weissbier is in line with published data.3
Figure 7. Intensity of isoxanthohumol (IX) and xanthohumol (X). Different levels of IX and X were observed across the different beers, shown here were the results for German Pilsner and a German Weissbier from the same brewery, as well as a dry-hoped IPA. The X/IX ratio in different beers showed a strong correlation with type of beer (Figure 8).
Figure 8. Observed peak intensities for isoxanthohumol (IX) and xanthohumol (X). The measured peak areas for Isoxanthohumul (blue) and Xanthohumol (green) are plotted on the left hand axis and show variability across the different beer types. The ratio of the two compounds (X/IX ratio in red, right hand axis) shows a clear difference for the Pale Ales and India Pale Ales. The data also reflects the differences of beers produced in different countries.
The concentrations of IX, X and X/IX from the beer samples are summarized in Figure 8. This data reflects different styles of beer making. It is obvious that the high X/IX ratio in Pale Ales and India Pale Ale (IPA) is caused by the late addition of hops during the boiling and fermentation process.4
Isoxanthohumol (IX), isocohumulone (ICH), and isohumulone (IH) were the 3 most abundant hop-derived bitter compounds detected in beer. ICH and IH and other iso-a-acids were present in their trans- and cis- form (Figure 9). The intensity ratio of trans/cis acids varied between 0.02 to 0.15 (American Lagers < European Pilsners < American Pal Ales and IPA).
Figure 9. Comparing the cis- and trans- ratios in bitter compounds. Intensity of cis- and trans-isocohumulone, isohumulone, cohumulone, and humulone in a Czech pilsner are shown, the trans / cis ratio was 0.06.
The measure of bitterness in beer is the IBU (International Bittering Units). A plot of the sum of the peak areas of IX, ICH and IH against the IBU for different beers is shown in Figure 10.
Figure 10. Correlation of the bitter compound intensities with the oublished international bitter units (IBU). Correlation of intensity of IX, ICH and IH against the published IBU for different beer styles (yellow: American lagers, orange: European pilsners, red: Pale Ales and India Pale Ales, brown: Bock, and black: Stout).
Beer profiling using statistical data analysis
Statistical data analysis, such as Principal Components Analysis (PCA), can be used to profile and compare different beverage samples. Figure 11 shows the PCA scores plot for 40 different beers produced using MarkerView Software. Beers of similar style group together. The location of a sample in the plot indicates a specific flavor or color (i.e. lighter beer vs. a more hoppy/bitter beer). The PCA loadings plot assists in finding characteristic markers (m/z-RT). Once these markers are found, the corresponding chemical can be identified using formula finding based on accurate mass MS and MS/MS followed by ChemSpider searching and MS/MS elucidation.
The b-acids lupulone and adlupulone were found in higher concentrations in darker beers, such as stout and black IPA, as the profile plot (bottom trace) in Figure 11 shows. Both compounds were identified using the described automated software tools in SCIEX OS Software (Figure 12).
Figure 11. Profiling differences between beer samples. Principle Components Analysis (PCA) using MarkerView Software was performed to profile and map different beer styles. This Scores plot (Top) shows how the beer styles cluster together. The loading plot (not shown here) assisted to find characteristic m/z-RT, selection of this feature can then be plotted across the different samples (bottom). The b-acids lupulone and adlupulone are plotted here and were found to be in higher concentrations in the darker beers.
Figure 12. Feature identification with SCIEX OS Software. Identification of lupulone and adlupulone in an Irish stout based on formula finding followed by ChemSpider searching and MS/MS elucidation
Summary
The SCIEX X500R QTOF System was used to analyze 40 different beer samples in positive polarity ESI using information dependent acquisition of MS and MS/MS spectra. Samples were processed using a targeted approach to profile 26 known hop-derived bitter compounds in SCIEX OS Software. Samples were also processed using PCA in MarkerView Software. This study shows that LC-MS/MS is a valuable tool to study flavor and aroma profile in food and beverage samples.
References
- J. P. Arnold (2005) Origin and History of Beer and Brewing: From Prehistoric Times to the Beginning of Brewing Science and Technology’ Alumni Association of the Wahl-Henius Institute (1911) Reprint Edition.
- R. Barth (2013) The Chemistry of Beer: The Science in the Suds. Wiley.
- D. Intelmann, G. Haseleu, and Th. Hofmann (2009) LC-MS/MS Quantitation of Hop-Derived Bitter Compounds in Beer Using the ECHO Technique. J. Agric. Food Chem. 57, 1172-1182
- J. F. Stevens, A. W. Alan, M. L. Deinzer (1999) Quantitative analysis of Xanthohumol and related prenylflavonoids in hops and beer by liquid chromatography tandem mass spectrometry. J. Chrom. A 832, 97-107.
- MarkerView™ Software 1.3.1 for statistical analysis of MS datasets. SCIEX technical note RUO-MKT-02-2775-A.
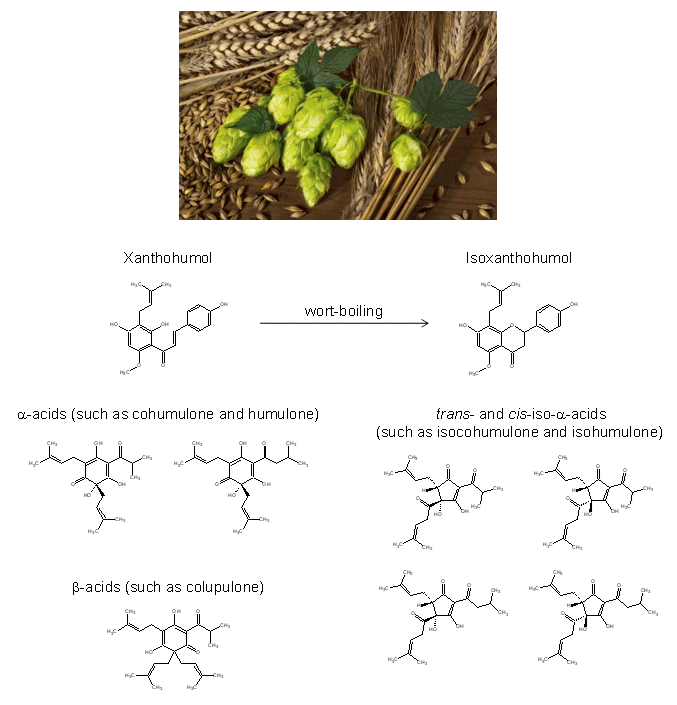 Click to enlarge
Click to enlarge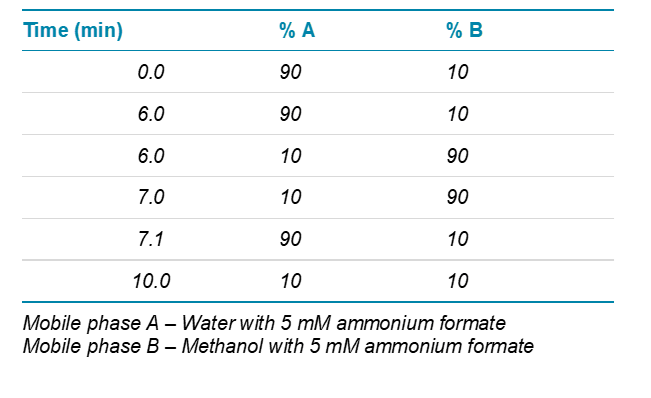 Click to enlarge
Click to enlarge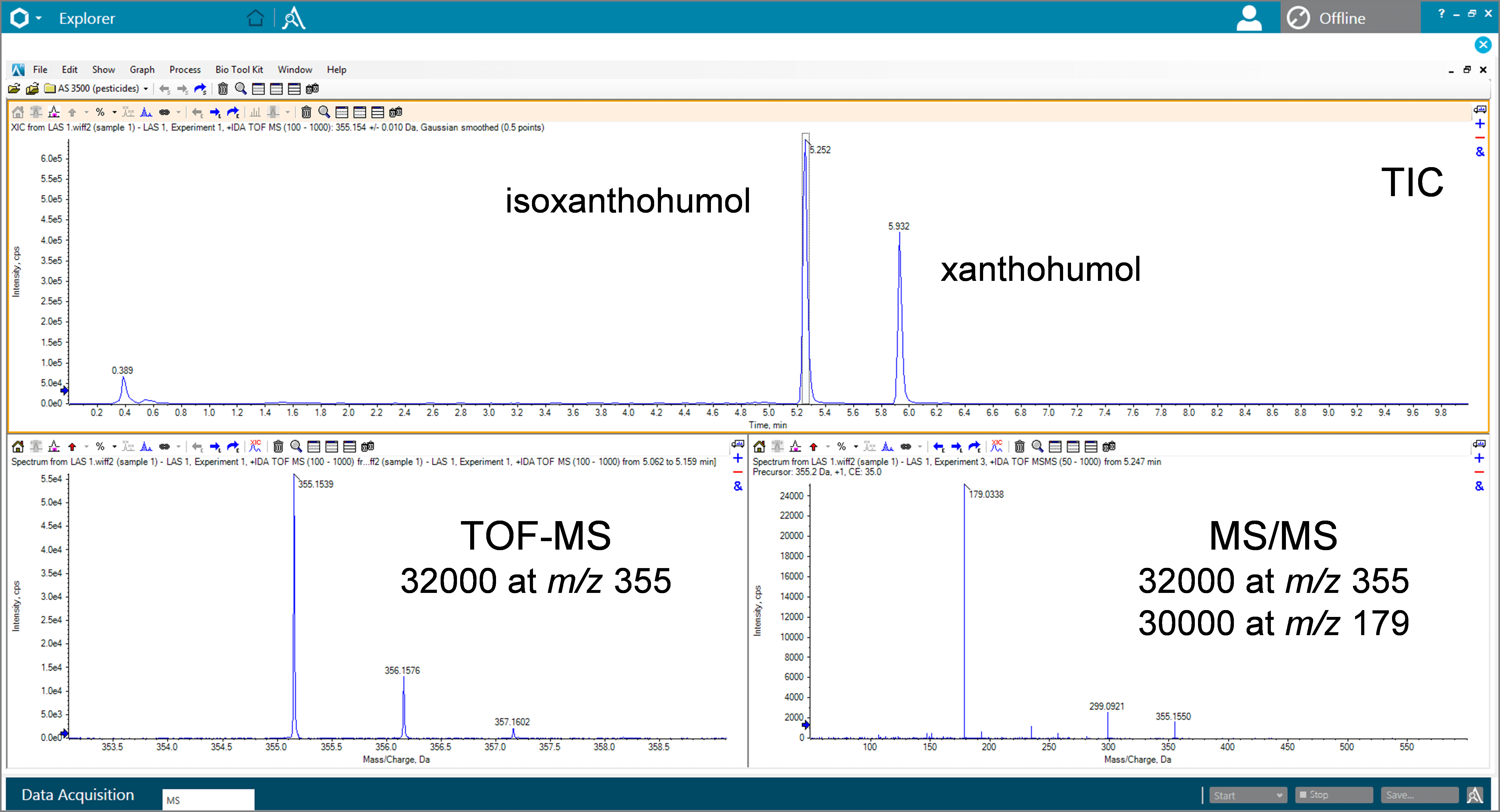 Click to enlarge
Click to enlarge Click to enlarge
Click to enlarge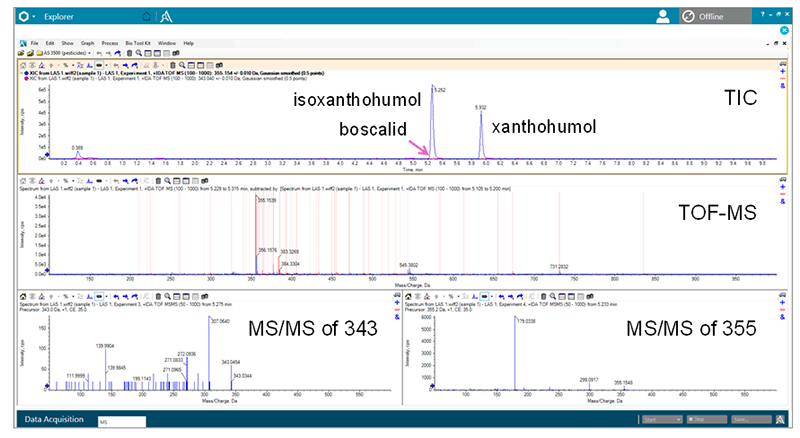 Click to enlarge
Click to enlarge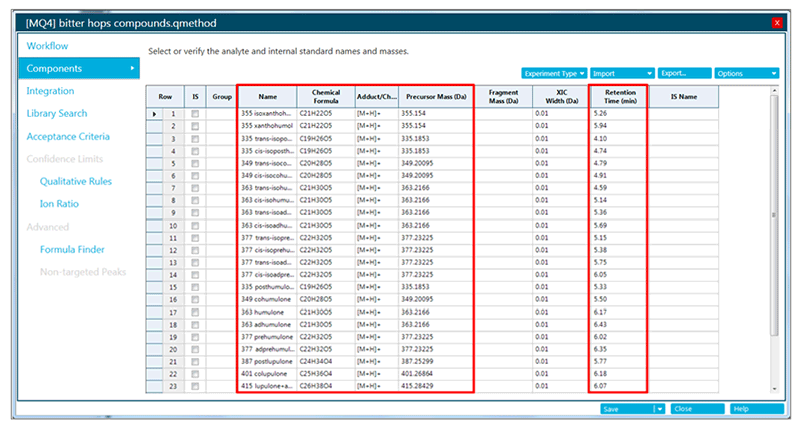 Click to enlarge
Click to enlarge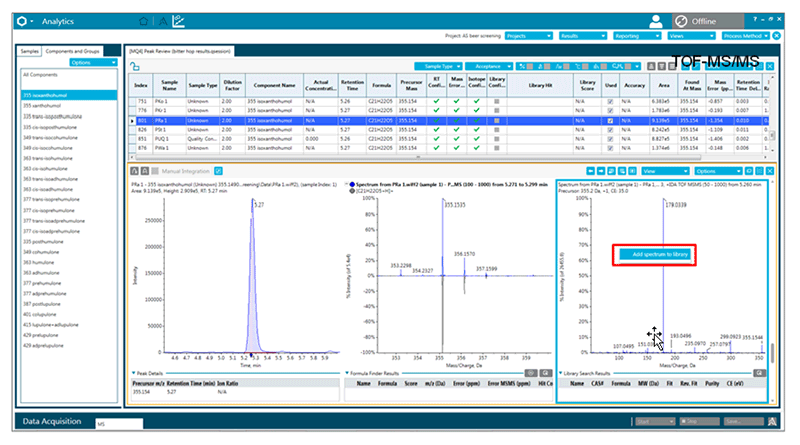 Click to enlarge
Click to enlarge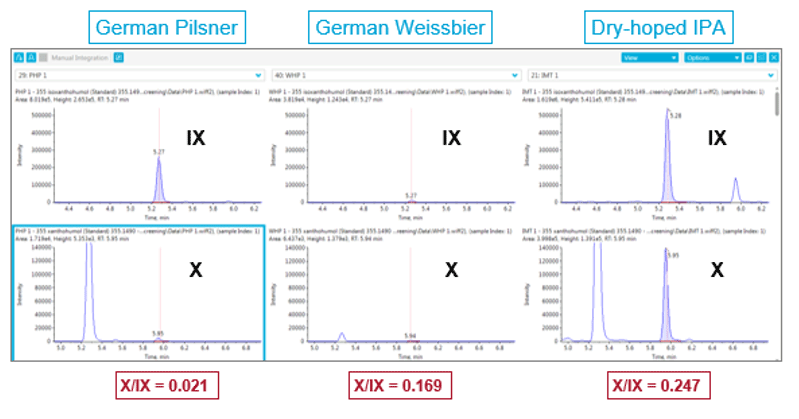 Click to enlarge
Click to enlarge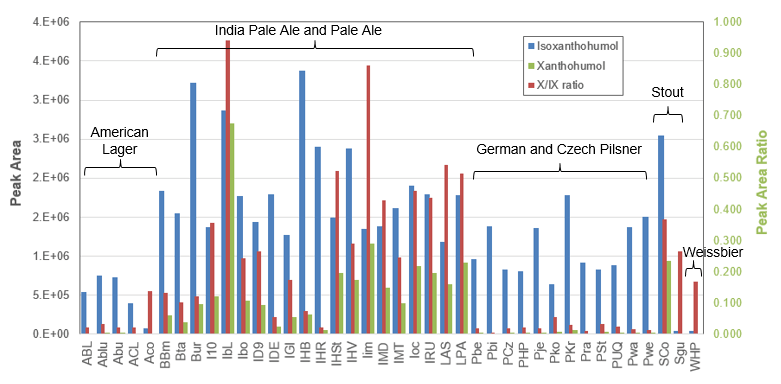 Click to enlarge
Click to enlarge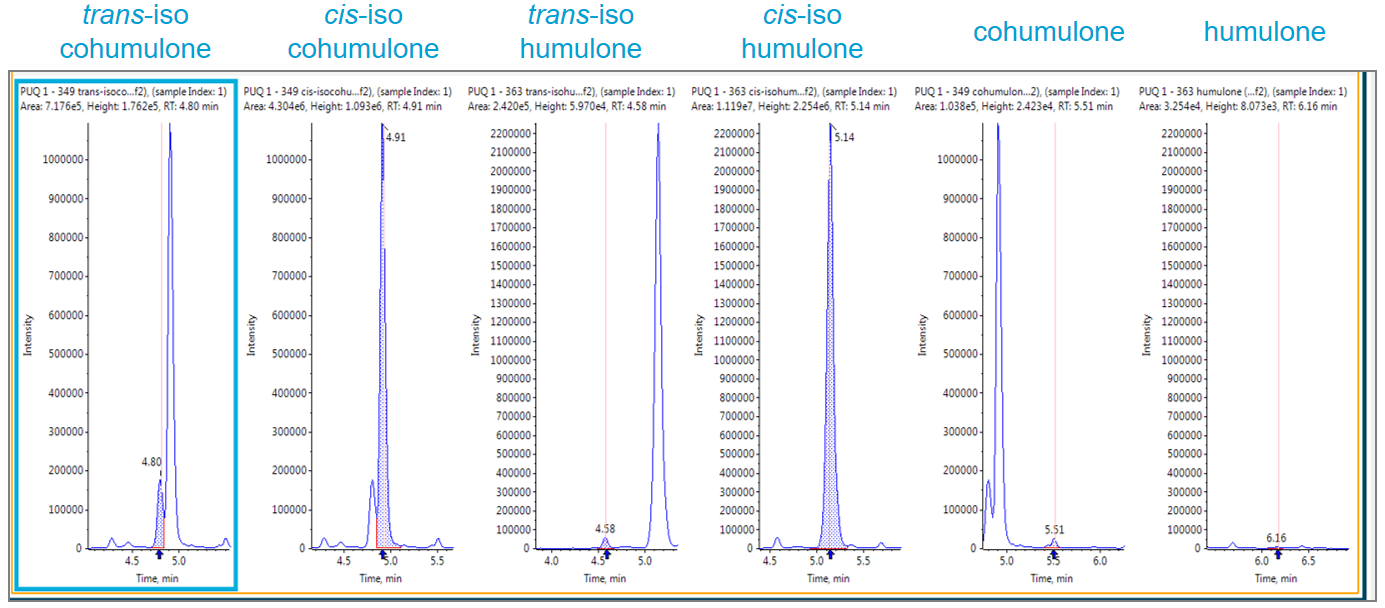 Click to enlarge
Click to enlarge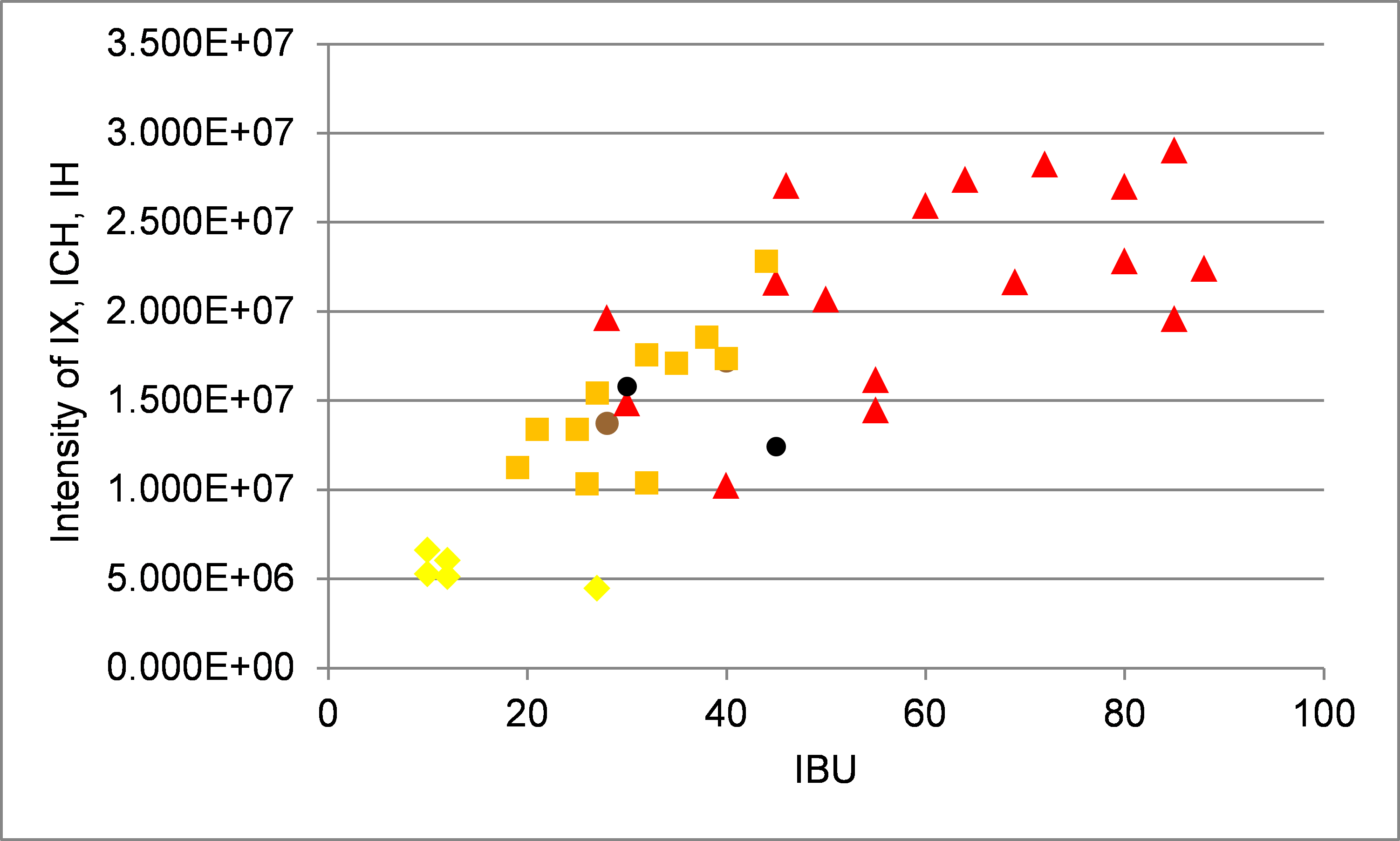 Click to enlarge
Click to enlarge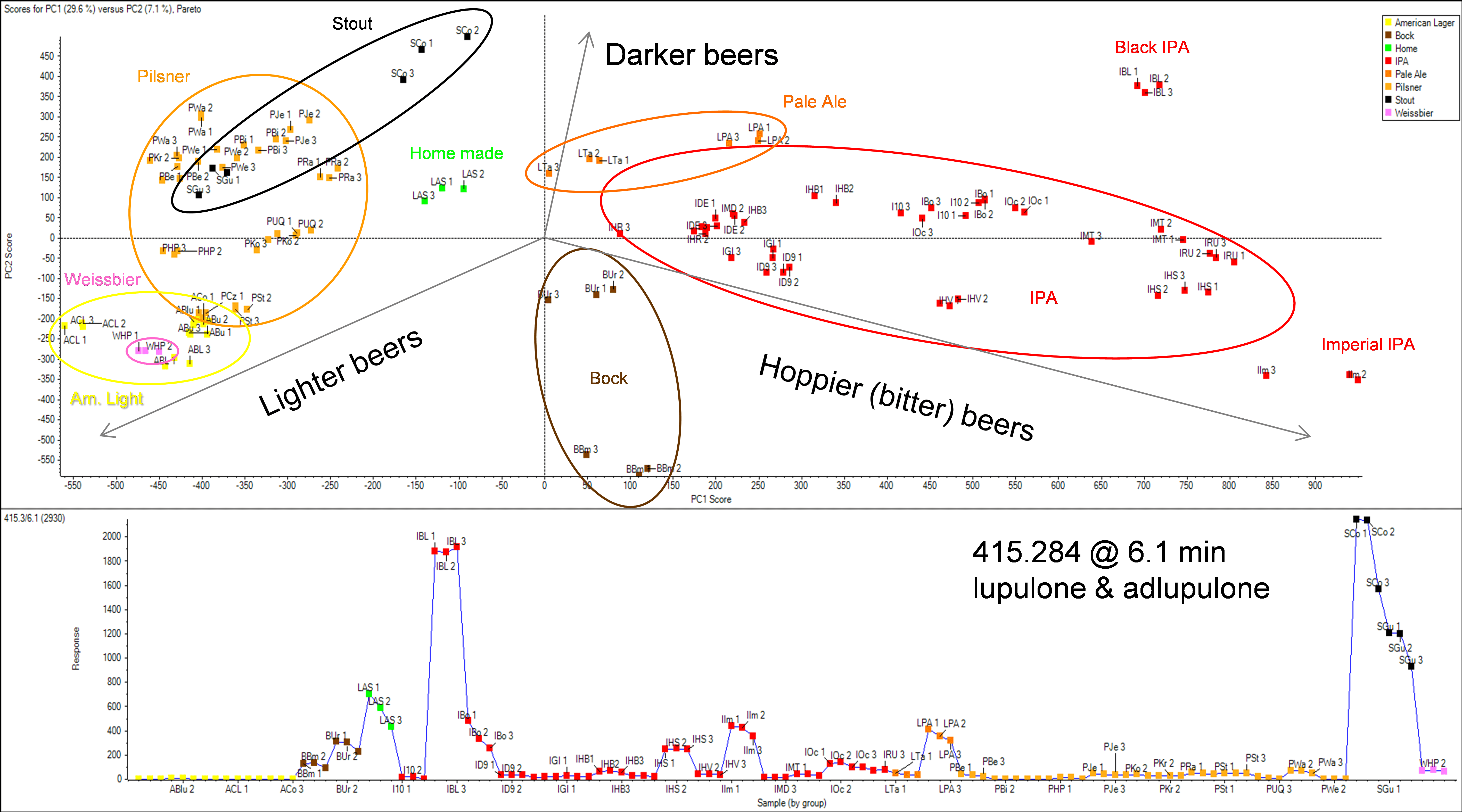 Click to enlarge
Click to enlarge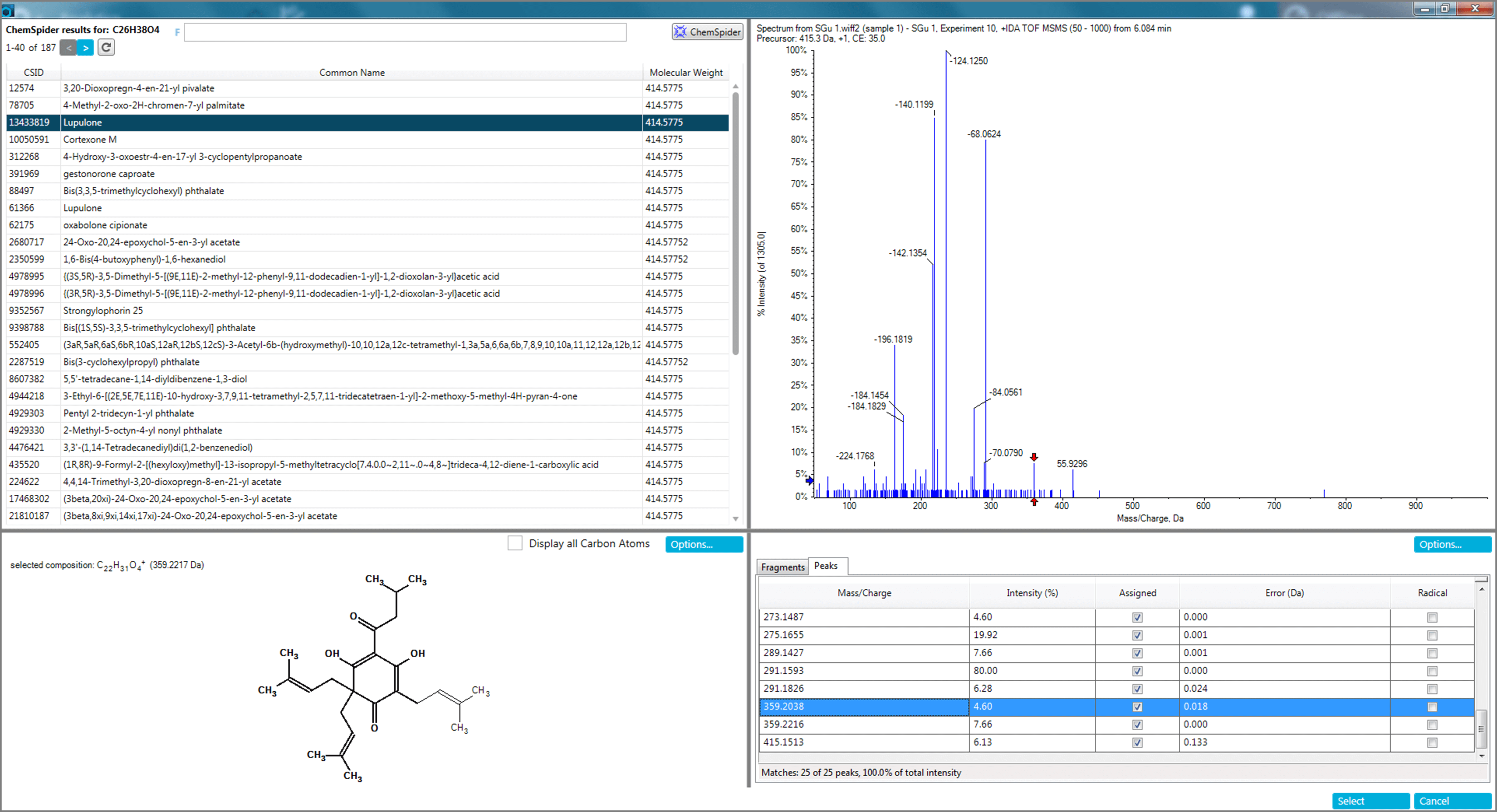 Click to enlarge
Click to enlarge