Optimize your workflows to reliably detect and monitor process-related impurities resulting from manufacturing of plasmids, cell-culture derived therapeutics and vaccines. Ensure the safety of your next-generation viral products with accurate and reproducible sizing of residual DNA and RNA and simultaneous quantitation.
Residual nucleic acid analysis

Overview
Residual nucleic acids require thorough assessment due to potential safety risks. Discover intuitive workflows for analyzing host cell DNA and RNA that provide meaningful answers based on high-quality results, and execute your product development and quality control processes with confidence.
Be empowered to tackle the analytical challenges of plasmids, vaccines and therapeutic proteins derived from bacterial, insect or mammalian cells.
Residual DNA analysis
Workflow
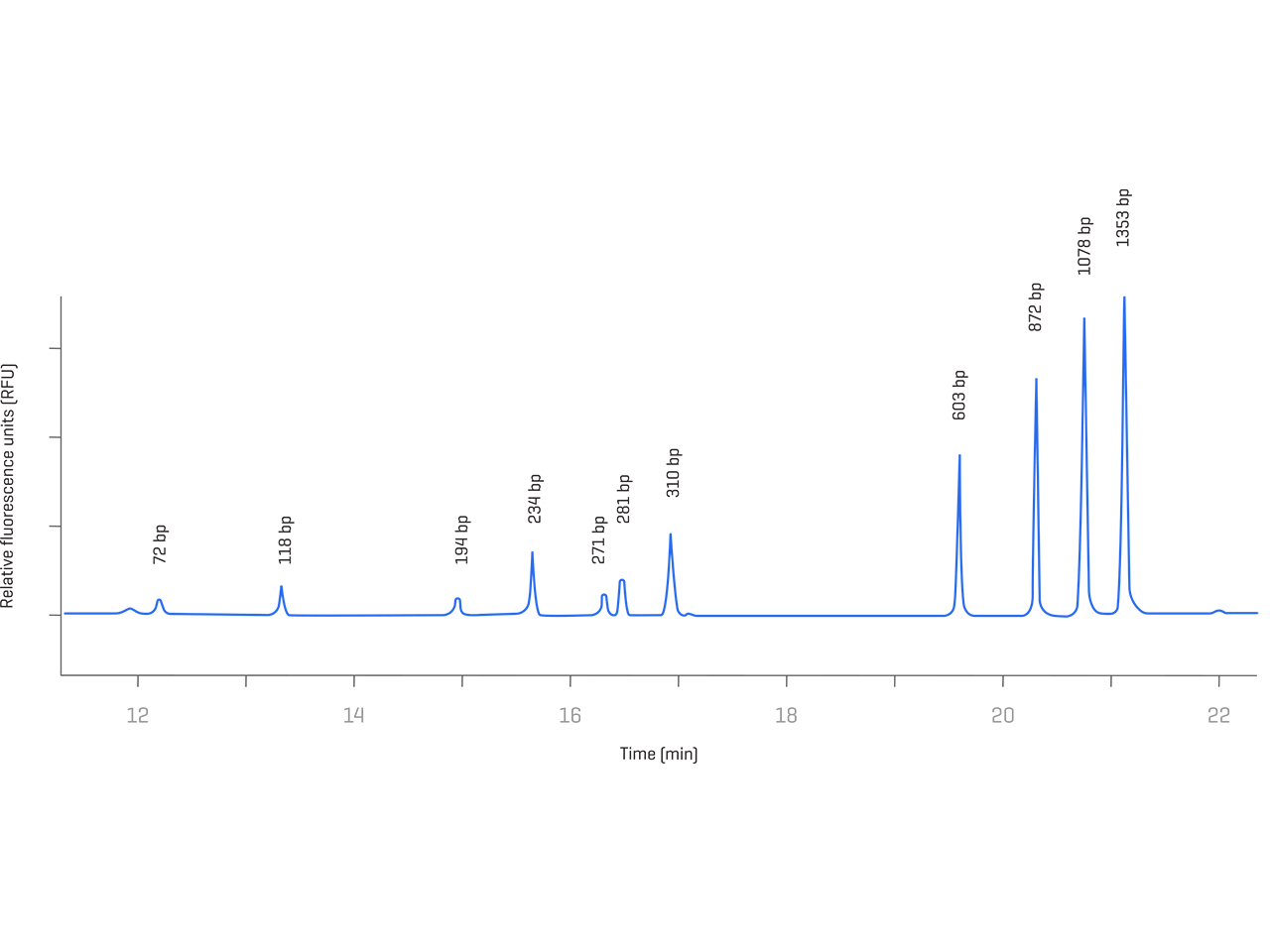
Host cell DNA poses a potential safety risk for cell culture-derived vaccines and therapeutic products. Reliable size determination and simultaneous quantitation of residual DNA are crucial to enable risk assessment and ensure product safety.
Be empowered to address safety concerns linked to residual DNA with high-quality solutions based on capillary gel electrophoresis. Understand your product changes and be confident in determining risks more easily and reproducibly.
- Determine risk based on the quantity and size of impurities
- Customize size ranges to your product needs
- Benefit from an intuitive and highly sensitive solution
- Increase confidence in DNA assessments with excellent quantitative performance
- Cover compliance needs through compatibility with the Empower Chromatography Data System (CDS)
Characterize therapeutic molecules with confidence with the kit-based system.
Featured resources
-
Webinar
Characterization of residual nucleic acids in cell culture-derived vaccines and therapeutics
Learn from the scientist Dr. Peter Holper about the need for residual nucleic acid analysis, and get insight into the benefits and drawbacks of different analytical techniques.
-
Publication
Size analysis of residual host cell DNA in cell culture-produced vaccines
Explore how the team at MedImmune addressed their challenges around residual host cell DNA and the associated potential safety concerns for cell culture-derived products. Learn how the scientists determined the quantity of residual DNA, assessed the size distribution and evaluated the performance of their optimized method.
-
Technical note
Size distribution analysis of residual host cell DNA fragments in lentivirus
Remove barriers to assessing residual nucleic acids and take control of process-related impurities with accurate and reliable workflows. Characterize sizes and quantities of residual host cell DNA for informed decision-making.
-
Technical note
Accurate analysis of double-stranded DNA over an extended size range
Take charge of analyzing double-stranded host cell DNA from 100 to 15,000 bp with excellent resolution and sensitivity across the entire size range. Determine product impurities with greater accuracy, repeatability and precision.
Residual RNA analysis
Workflow
Residual host cell RNA can affect product quality and pose risks, such as eliciting undesired immune responses.
Take control of process-related nucleic acid impurities with accurate, reliable and high-quality data. Assess size distribution over a wide range and determine associated quantities with confidence. Be empowered to make informed decisions for risk assessments with turnkey solutions that meet your needs.
- Determine risk based on the quantity and size of impurities
- Avoid the sequence bias associated with methods based on polymerase chain reaction (PCR)
- Benefit from an intuitive and highly sensitive solution
- Increase confidence in impurity assessments with excellent quantitative performance
- Cover compliance needs through compatibility with the Empower CDS
Residual RNA analysis
Purpose-built for the biopharmaceutical scientist for efficiency and quality, enabling multiple samples to be run in parallel.
Purity and integrity analysis of RNA therapeutics, vaccines and single stranded oligos.
Pre-assembled bare-fused silica 8-capillary cartridge for the BioPhase 8800 system - 8 x 30 cm. (P/N 5080121)
Residual RNA analysis
Characterize therapeutic molecules with confidence with the kit-based system.
Purity and integrity analysis of RNA therapeutics, vaccines and single stranded oligos.
Pre-assembled bare-fused silica capillary cartridge – 30 cm for the PA 800 Plus system.
Featured resources
-
Webinar
Characterization of residual nucleic acids in cell culture-derived vaccines and therapeutics
Learn from the scientist Dr. Peter Holper about the need for residual nucleic acid analysis, and get insight into the benefits and drawbacks of different analytical techniques.
-
Technical note
Comprehensive method development for a wide size range of RNA
Harness the power to assess RNA up to 9,000 nucleotides with excellent resolution and sensitivity across the entire size range.
All resources
-
Webinar
Characterization of residual nucleic acids in cell culture-derived vaccines and therapeutics
Learn from the scientist Dr. Peter Holper about the need for residual nucleic acid analysis, and get insight into the benefits and drawbacks of different analytical techniques.
-
Publication
Size analysis of residual host cell DNA in cell culture-produced vaccines
Explore how the team at MedImmune addressed their challenges around residual host cell DNA and the associated potential safety concerns for cell culture-derived products. Learn how the scientists determined the quantity of residual DNA, assessed the size distribution and evaluated the performance of their optimized method.
-
Technical note
Size distribution analysis of residual host cell DNA fragments in lentivirus
Remove barriers to assessing residual nucleic acids and take control of process-related impurities with accurate and reliable workflows. Characterize sizes and quantities of residual host cell DNA for informed decision-making.
-
Technical note
Accurate analysis of double-stranded DNA over an extended size range
Take charge of analyzing double-stranded host cell DNA from 100 to 15,000 bp with excellent resolution and sensitivity across the entire size range. Determine product impurities with greater accuracy, repeatability and precision.
-
Technical note
Genome integrity and nucleic acid impurity analysis of adeno-associated viruses (AAVs)
Reclaim your time by simultaneously assessing viral vector genome integrity and purity with high resolution over an extended size range. Determine impurity quantity and size information for confident decision-making.
-
Technical note
Comprehensive method development for a wide size range of RNA
Harness the power to assess RNA up to 9,000 nucleotides with excellent resolution and sensitivity across the entire size range.
Associated applications
Optimize your processes with solutions that overcome the analytical challenges of viral vector-based drugs. Take control of full and empty ratios and protein and genome profiles. Break down the boundaries of protein and post-translational modification (PTM) characterization with dedicated high-resolution workflows offering unprecedented depth.
Advance your projects with purpose-built solutions for the analysis of plasmid DNA (pDNA) and double-stranded DNA (dsDNA) to support the development of your messenger RNA (mRNA), viral vectors, recombinant proteins and other new modalities.