Experimental details
Target analytes and solutions: A total of 49 drugs and 18 deuterated internal standards were purchased from Cerilliant Corporation (Round Rock, TX). Two solutions were prepared in methanol: a standard mixture containing the 49 target analytes and an internal standard mixture containing the 18 deuterated internal standards. Table 1 lists the name, the calibration range, linear correlation value (R2), and LLOQ, as well as the accuracy and precision reported at the LLOQ for each of the 49 target analytes used in this panel.
Calibrator preparation: Eight levels of calibrators ranging from 10 µg/mL to 1 pg/mL were prepared in methanol. A 10 ng/mL IS standard stock solution was prepared in MeOH/water (20:80, v/v) for sample reconstitution prior to injection.
Sample preparation: 10 µL of each calibrator solution was spiked into 90 µL of human whole blood. Each spiked human whole blood sample was extracted by using a protein precipitation procedure. In short, 900 µL of methanol/acetonitrile (50:50, v/v) were added into each of the spiked human whole blood samples and vortexed for 1 min then followed by 3 min sonication and another 1 min of vortex mixing. The samples were then centrifuged for 5 min at 8,000 rpm. The supernatant was transferred out to a glass tube and completely dried down under nitrogen gas. The residues were reconstituted with 500 µL of a 10 ng/mL IS standard stock solution in methanol/water (20:80, v/v). The protein precipitation procedure is shown in Figure 2.
Liquid chromatography: HPLC separation was performed on an ExionLC™ System using a Phenomenex Kinetex Phenyl-Hexyl column (50 × 2.1 mm, 2.6µm, 00B-4495-E0). The separation conditions were identical to those previously described in a technical note.2 Mobile phases were ammonium formate in water (MPA) and formic acid in methanol (MPB). The injection volume was 5 µL and the LC runtime was 6.5 min.
Mass spectrometry: A SCIEX Triple Quad 7500 System equipped with an OptiFlow Pro Ion Source with an analytical probe and E Lens™ Technology was used. The ionization source was operated in electrospray ionization (ESI) mode in both positive and negative polarities. A single acquisition method consisting of 134 MRM transitions (98 for the drugs and 36 for the internal standards) was created using the Scheduled MRM Algorithm Pro in SCIEX OS Software 2.0. Two MRM transitions were monitored for each of the targeted drugs and each sample was injected in triplicate to build a data analysis processing method.
Data analysis: Data processing was performed using SCIEX OS Software. Detection and integration of the peaks from the background was achieved within the viewing window using the AutoPeak algorithm. Quantitative analysis was performed in the Analytics module of the software where calibration curves, concentration calculations, assay precision and accuracy statistics were generated.
Method development and optimization
A diluted, neat standard mixture containing the 49 target analytes was used for initial method development. The Scheduled MRM Algorithm Pro in SCIEX OS Software was used to optimize data sampling across each peak while maintaining optimal dwell times for each MRM transition to ensure reliable integration, quantification and confirmation of the peak for each target analyte. In addition, fast polarity switching was used to provide maximum analyte coverage. Most MRM transitions had 15 or more data points across each of the LC peaks, with 10 being the minimum number of data points across each peak for each of the 49 target analytes used in this study. Figure 3 shows the elution profile for the 49 targeted drugs resulting from the optimized data acquisition method.
Optimized detection method enables accurate and reliable drug quantification
Control human whole blood samples spiked with the 49 target analytes were prepared at concentrations ranging from 1 µg/mL down to 0.1 pg/mL. Detection and integration of the peaks was performed automatically using the AutoPeak Algorithm in the Analytics module of SCIEX OS Software. Analyte concentration and ion ratio were calculated automatically in the software.
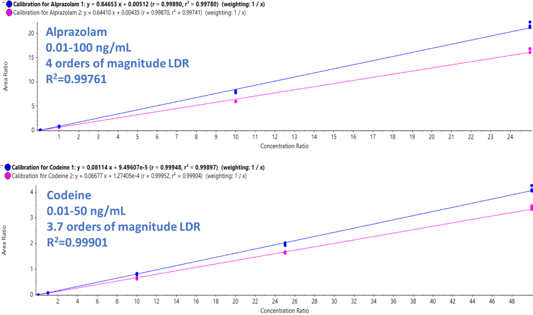
The potential for drastic variation in detected drug levels in toxicology case samples necessitates the use of an instrument with high range of linear response. Figure 1 shows calibration curves for two of the drugs targeted in this study. Concentration range of 4 (from 0.01-100 ng/mL) and 3.7 (from 0.01-50 ng/mL) orders linear dynamic range was demonstrated for alprazolam and codeine, respectively. Excellent linearity was observed across the concentration ranges analyzed with R2 average values of 0.99761 and 0.99901 for alprazolam and codeine, respectively. Similar trends were observed for the other analytes used in this study.
OptiFlow Pro Ion Source and E Lens Technology leads to enhanced sensitivity
Developing robust workflows that can deliver high levels of sensitivity is critical to any toxicology laboratory needing to quantify a wide concentration of drugs extracted from biological specimens. To this end, the sensitivity of the SCIEX Triple Quad 7500 System was assessed by determining the lower limit of quantification (LLOQ) values for the 49 targeted drugs in the panel. LLOQ values were determined as the lowest concentration calibration level fitting the following standard performance requirements: signal-to-noise ratio (S/N) of at least 10, calculated concentration accuracy within 20% of 100%, precision (%bias) below 25%, and falling on a linear calibration curve with an R2 value of at least 0.98.
Figure 4 shows the extracted ion chromatogram (XIC) traces and resulting calibration curves of the two MRM transitions monitored for 7-hydroxymitragynine (Figure 4A) and acetyl fentanyl (Figure 4B). The two series of XIC traces for both the quantifier and qualifier ions of each of the two detected drugs showed a high level of sensitivity and precision across the calibration series for concentrations ranging from 0.1 to 100 ng/mL for 7-hydroxymitragynine and 0.02-50 ng/mL for acetyl fentanyl, respectively. Six levels of calibrators were used to determine the ion ratio criteria for the quantifier and qualifier ions of these two targeted drugs. The results demonstrated excellent correlation of the generated regression curves covering concentration ranges far exceeding typical bioanalytical requirements.
Table 1 summarizes the statistical results obtained for the 49 analytes spiked in human whole blood. The table includes calibration range, linear correlation coefficient (R2 Value), and LLOQ, as well as the accuracy and precision for each of the two MRM transitions monitored for each drug. Overall, the assay showed excellent reproducibility, precision, accuracy, and linearity, proving the robustness and performance of the developed method.
Figure 5 shows the statistical results and the calibration curves resulting from the peak area integration of dihydrocodeine (Figure 5A) and noroxycodone (Figure 5B) from 0.5 to 100 ng/mL. Excellent linearity, reproducibility, accuracy and precision was observed across the six calibration levels covering the concentration range. The assay showed excellent precision and accuracy, and the averaged R2 values for the quantifier and qualifier ions were 0.99571 and 0.99323, respectively. Full quantification was achieved with SCIEX OS Software, designed for quick, intuitive and streamlined data processing with accurate and reliable results.
Conclusions
An optimized and sensitive method for the detection of a panel of 49 drugs in human whole blood is described using the SCIEX Triple Quad 7500 System. The use of the Scheduled MRM Algorithm Pro in SCIEX OS Software enabled optimization of data sampling. The addition of new hardware features, the OptiFlow Pro Ion Source, the D Jet Ion Guide and the E Lens, provided sensitive quantification of all the targeted drugs in the sub ng/mL range, with some down to the pg/mL levels, while maintaining linearity, precision and accuracy of measurement. This exceptional sensitivity was achieved without any sacrifice or compromise to data quality, as demonstrated by the excellent precision and accuracy observed at the LLOQ. Overall, the combination of the features on the SCIEX Triple Quad 7500 System results in unparalleled sensitivity improvement for the suite of drugs targeted in this study.

 Click to enlarge
Click to enlarge Click to enlarge
Click to enlarge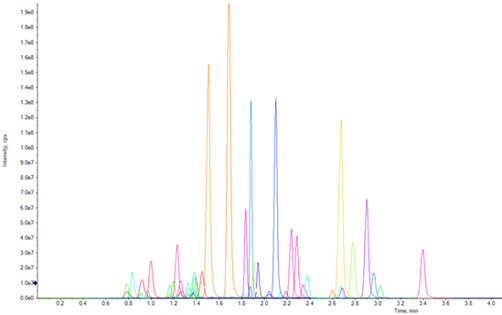 Click to enlarge
Click to enlarge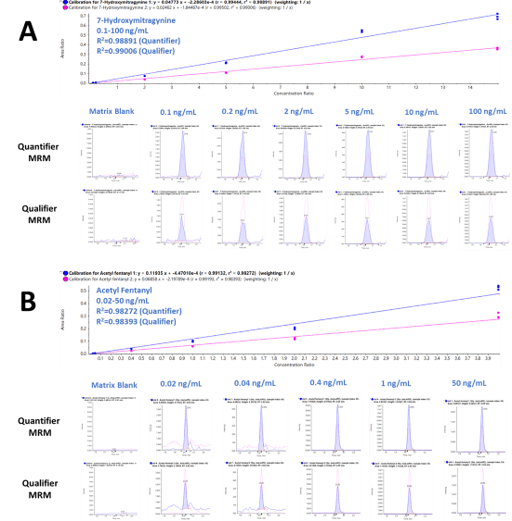 Click to enlarge
Click to enlarge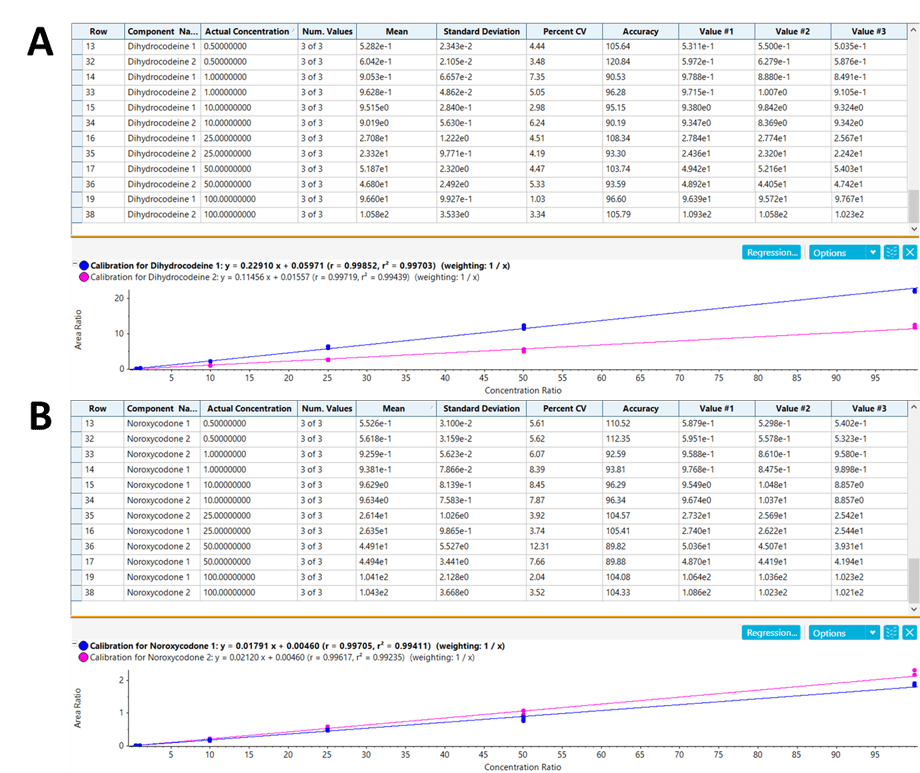 Click to enlarge
Click to enlarge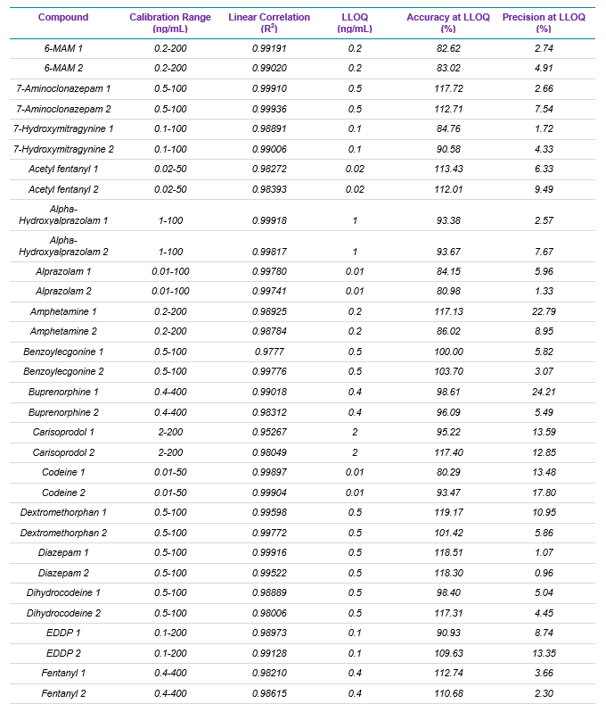 Click to enlarge
Click to enlarge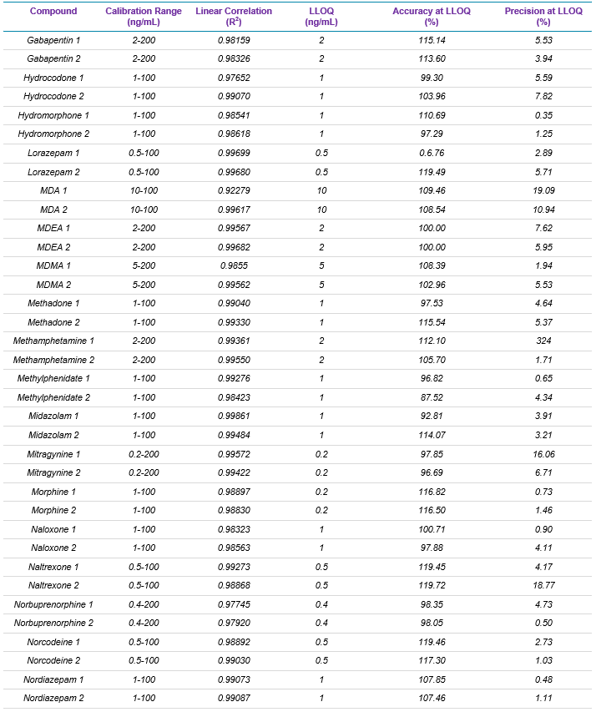 Click to enlarge
Click to enlarge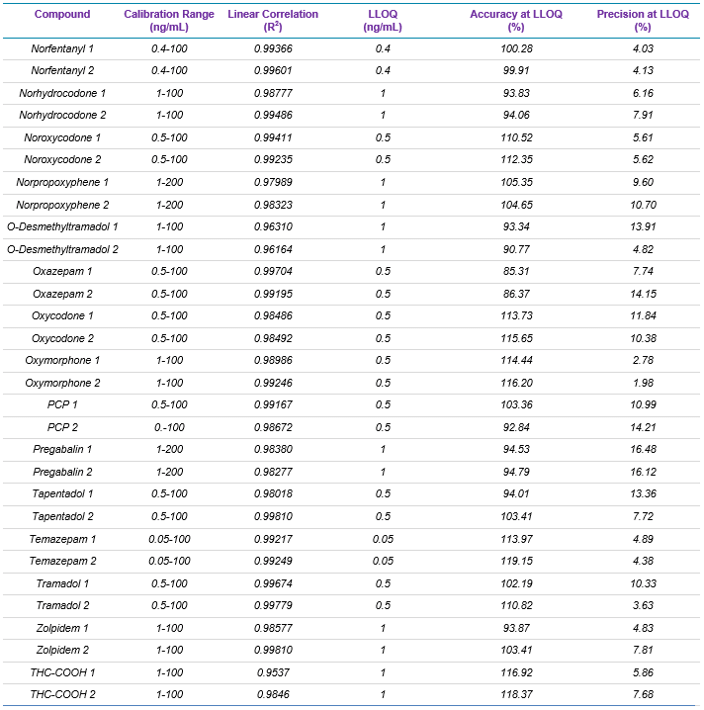 Click to enlarge
Click to enlarge