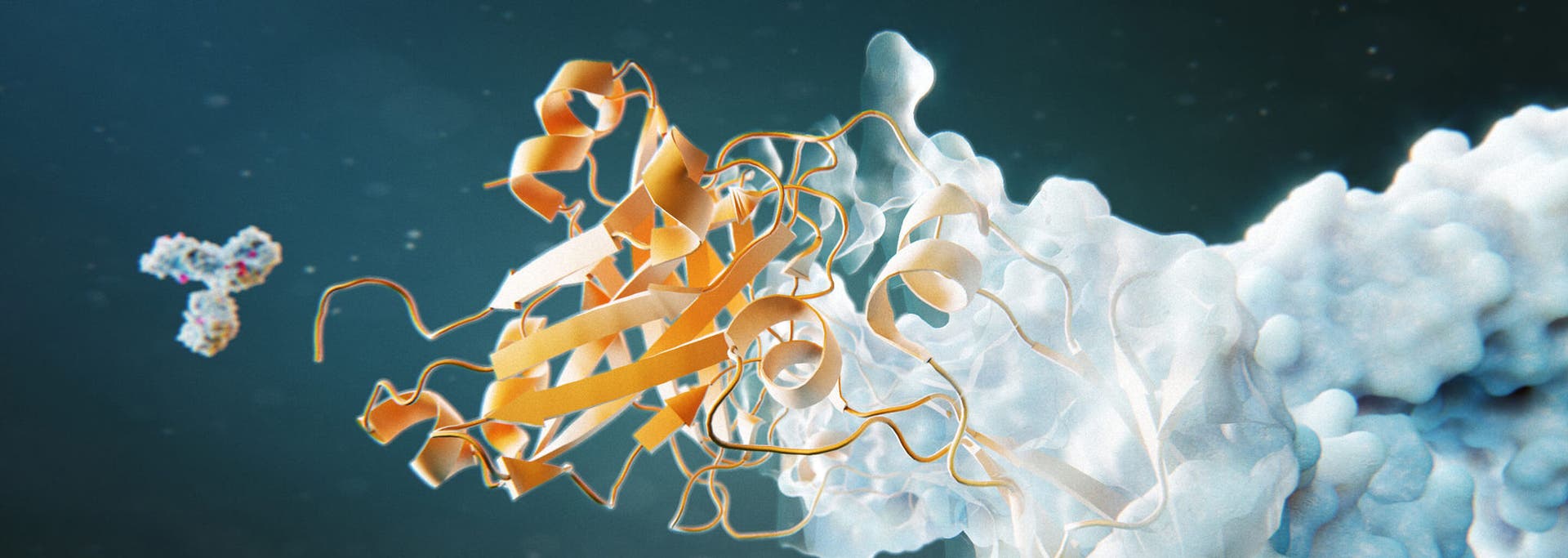
The requirements of peptide mapping analyses change during the drug development process. In early development, full characterization and determining the appropriate product quality attributes (PQAs) are key and depend on extremely complex data processing and analysis. In late development, throughput is a priority to quickly process samples and analyze PQAs. Over the course of biotherapeutic production, continuous monitoring of PTMs and chemical variations is critical to maintain overall product safety and efficacy.
Address the need for complete sequence coverage, comprehensive determination of PTMs and reliable high-throughput monitoring assays with LC-MS solutions designed to address the challenges of peptide mapping at all stages of the therapeutic development pipeline.