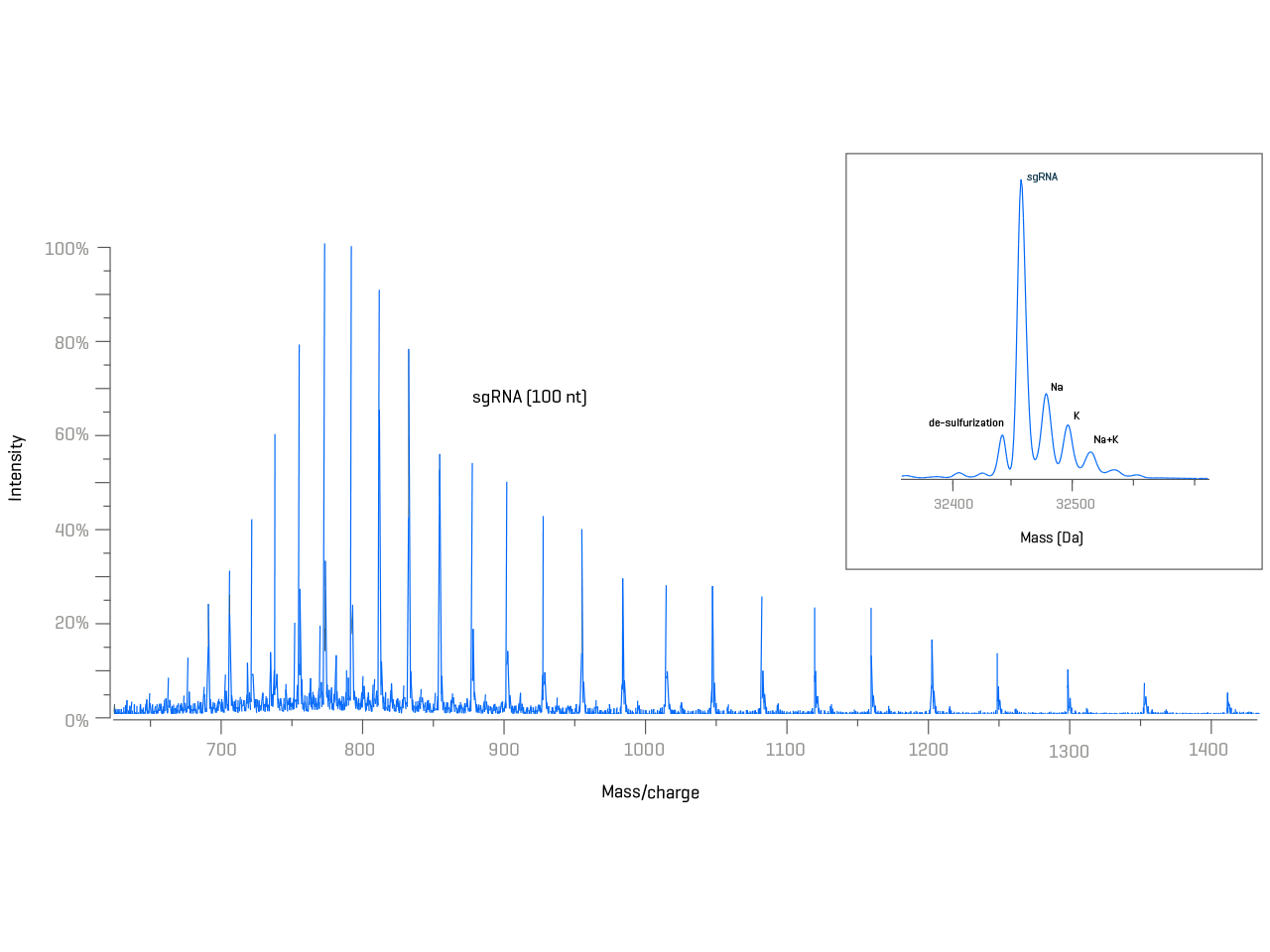
Work in pursuit of a higher level of quality for characterization of synthetic oligonucleotides—such as single guide RNA (sgRNA) or prime editing guide RNA (pegRNA)—used for CRISPR/Cas9 gene editing applications, along with antisense oligonucleotides (ASOs), small interfering RNA (siRNA) and aptamer therapeutics.
Synthetic oligonucleotide analysis

Explore the latest and trending topics
-
Technical note
Sizing and purity analysis of prime editing guide RNA (pegRNA)
Experience the impact of overcoming secondary structure challenges presented by large-sized oligonucleotides for gene editing. Learn how a high-resolution solution enables inutitve and accurate sizing and purity analysis of complex synthetic prime editing guide RNA (pegRNA).
-
Technical note
Visualize intact oligonucleotide data for rapid sample quality and impurity assessment
Discover a user friendly workflow for rapid and confident identification, impurity assessment, and characterization of synthetic oligonucleotides, with software that provides flexible tools for sequence creation, deconvolution, and mass mapping.
Overview
Synthetic nucleotide analysis is a complex pursuit. Nucleic acid-based drugs combine the learnings from nature’s mechanisms with sophisticated technology. The diverse landscape of structural adaptions for bases and backbones has the potential to treat diseases with unprecedented specificity, but also increases the challenges for analyzing the synthetic oligonucelotide.
Move beyond boundaries for sequence confirmation, impurity ID, sizing and purity analysis and more with cutting-edge analytical solutions.
High-quality results for therapeutic oligos (ASO, siRNA, aptamers) and CRISPR/Cas9-related nucleic acids (gRNA, sgRNA, pegRNA) help you advance medicine.
Impurity ID
Workflow
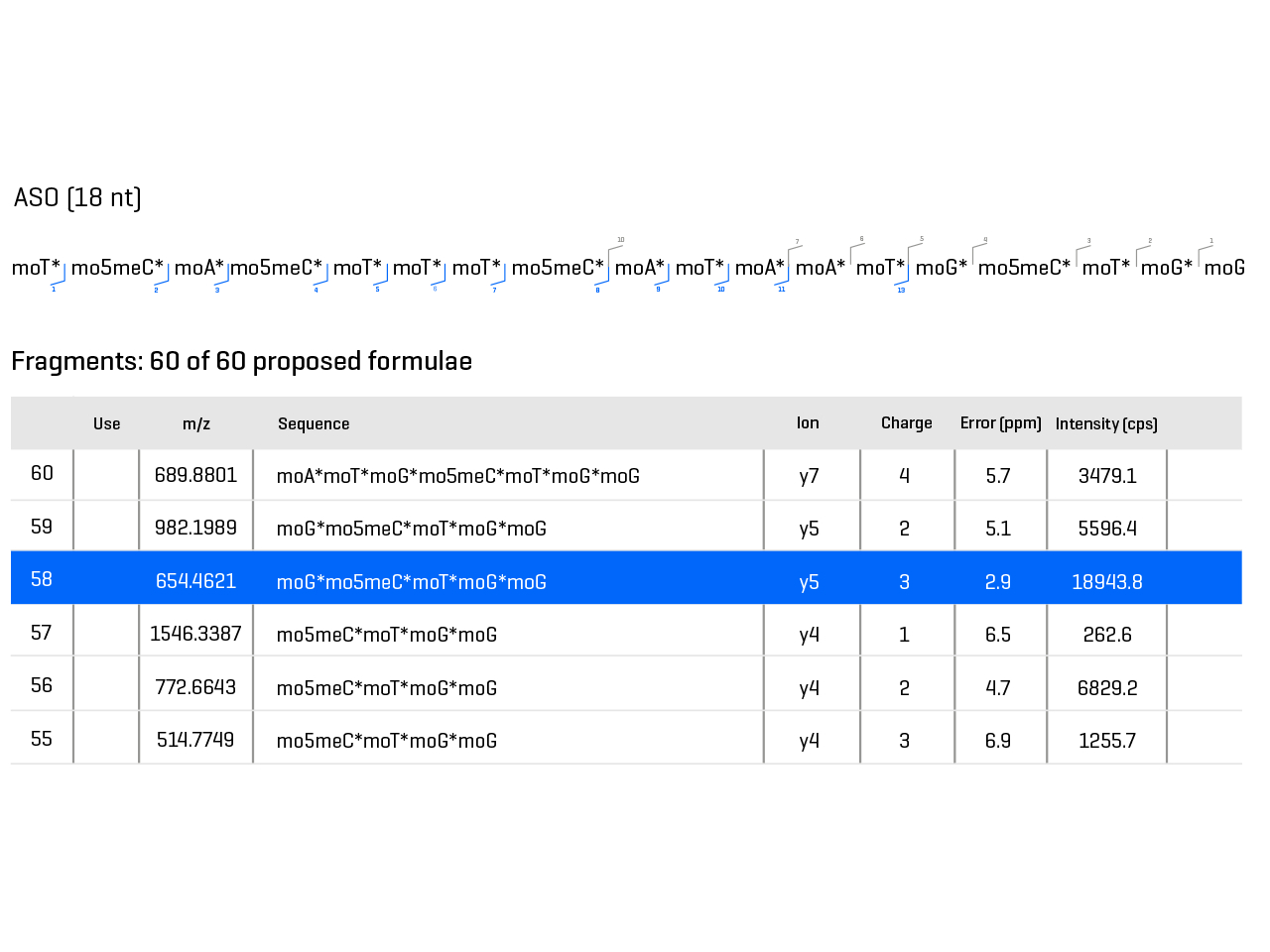
Due to their stepwise synthesis, therapeutic oligonucleotides can contain a number of impurities with highly similar properties. These frequently extensively modified modalities show very complex fragmentation data that is difficult to interpret.
Take control of identity confirmation for your product and mitigate risks of impurities while keeping salt-adducts down. Don’t let base and backbone modifications hold you back. Determine the molecular weight, sequence and quanities of the active pharmaceutical ingredient (API) as well as impurities with customizable workflows.
- Identify API and impurities with intuitive processing software allows full flexibility for customized bases and phospho-backbones
- Achieve high sensitivity and superior de-clustering of salt adducts with state-of the art source design
- Gain trust in your sequence confirmation with high MS/MS data quality
- Stay flexible for future quantitation challenges with deconvolution-based quantitation
- Keep your team moving with intuitive acquisition software
Impurity ID
Suited for:
- Identfication and quantitation of impurities
- Robust, analytical flow setup
Reproducibility, reliability and carryover performance to match your quantitative workflows. Dependability you can count on, from injection to injection and batch after batch.
A purpose-built QTOF system, designed specifically to accelerate everyday biologics characterization.
Determine impurities and biotransformations for a wide variety of modalities.
Unleash the analytical power of the next-generation software platform for data acquisition and processing.
Impurity ID
Suited for:
- Highly sensitive impurity ID and quantitation with superior linear dynamic range (LDR)
- Meeting flexibility needs to perform a range of additional workflows
Reproducibility, reliability and carryover performance to match your quantitative workflows. Dependability you can count on, from injection to injection and batch after batch.
A high-resolution mass spectrometry solution that combines powerful MS/MS sensitivity, fragmentation technology and a step-change in data independent acquisition.
Determine impurities and biotransformations for a wide variety of modalities.
Unleash the analytical power of the next-generation software platform for data acquisition and processing.
Featured resources
-
Technical note
Characterization of oligonucleotides and related impurities to support the development of nucleic acid-based therapeutics
Remove barriers to the detection of impurities in your therapeutic oligonucleotide drugs. Explore how to achieve impurity ID with full sequence coverage for highly modified ASOs present at levels as low as 0.3%.
-
Technical note
Molecular weight determination and impurity analysis of a 70-mer oligonucleotide
Imagine taking control of your CRISPR/Cas9 gene-editing systems by ensuring sgRNA integrity and purity. Be empowered to take on the challenge of future medicine development with accurate mass solutions that provide meaningful answers.
Sizing and purity analysis
Workflow
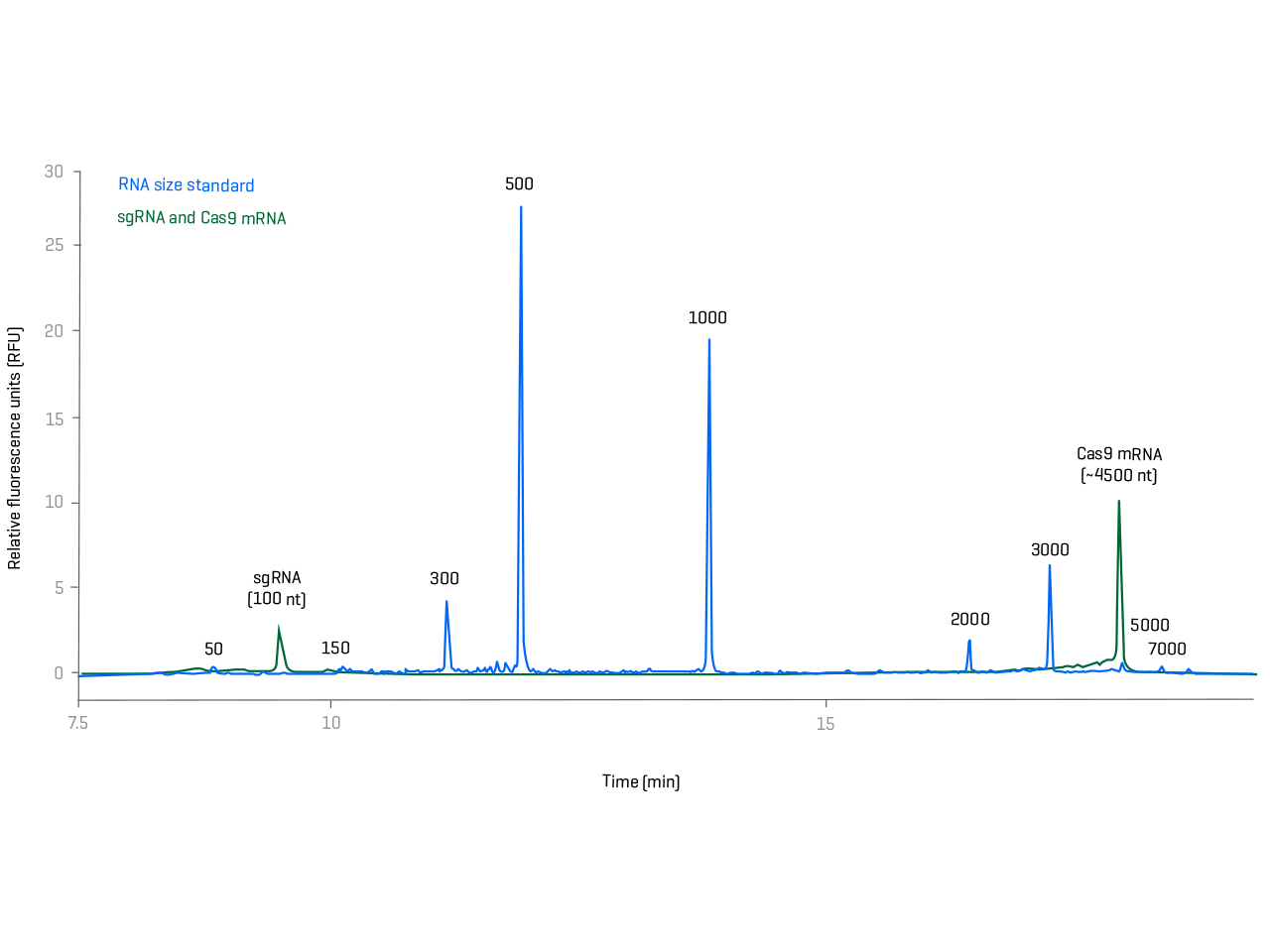
The latest gene-editing approaches require overcoming new analytical challenges. Confirmation of the size, integrity and purity of large synthetic guiding RNAs requires the highest data quality over a wide size range.
Move past analytical challenges with high separation quality, sensitivity and superior reproducibility. Reclaim your time with intuitive software and kit-based workflows that are compatible with compliant data systems.
- High-resolution measurement of synthetic RNAs over a wide size range
- High repeatability and minimized secondary structure formation with stable temperature control of the capillary and cartridge
- High-sensitivity detection of analytes and low-abundance impurities
- Easy assay transfer to regulated environments due to compatibility with the Empower Chromatography Data System (CDS)
Sizing and purity analysis
Suited for:
- High-quality resolution and sizing of RNAs and impurities
- Medium to large sample batches
Purpose-built for the biopharmaceutical scientist for efficiency and quality, enabling multiple samples to be run in parallel.
Purity and integrity analysis of RNA therapeutics, vaccines and single stranded oligos.
Pre-assembled bare-fused silica 8-capillary cartridge for the BioPhase 8800 system - 8 x 30 cm. (P/N 5080121)
Featured resources
-
Technical note
High-resolution size and purity determination of sgRNA and Cas9 mRNA in a single analysis
Learn about an intuitive kit-based solution for the characterization of mRNA integrity and purity. Break through boundaries with workflows that provide the highest resolution for sgRNA and mRNA in a single analysis.
-
Technical note
Compliant-ready, streamlined data management for Cas9 mRNA analyses with the Empower CDS
Set your own schedule for mRNA analyses by streamlining your CE data management with the Empower CDS.
-
Webinar
Empowering next-generation therapy development with streamlined data integration
Discover how to save time by streamlining your CE data management with the Empower CDS.
Sizing and purity analysis
Suited for:
- High-quality resolution and sizing of RNAs and impurities
- Smaller sample sets
Characterize therapeutic molecules with confidence with the kit-based system.
Purity and integrity analysis of RNA therapeutics, vaccines and single stranded oligos.
Pre-assembled bare-fused silica capillary cartridge – 30 cm for the PA 800 Plus system.
Sizing and purity of large RNAs (pegRNA)
Workflow
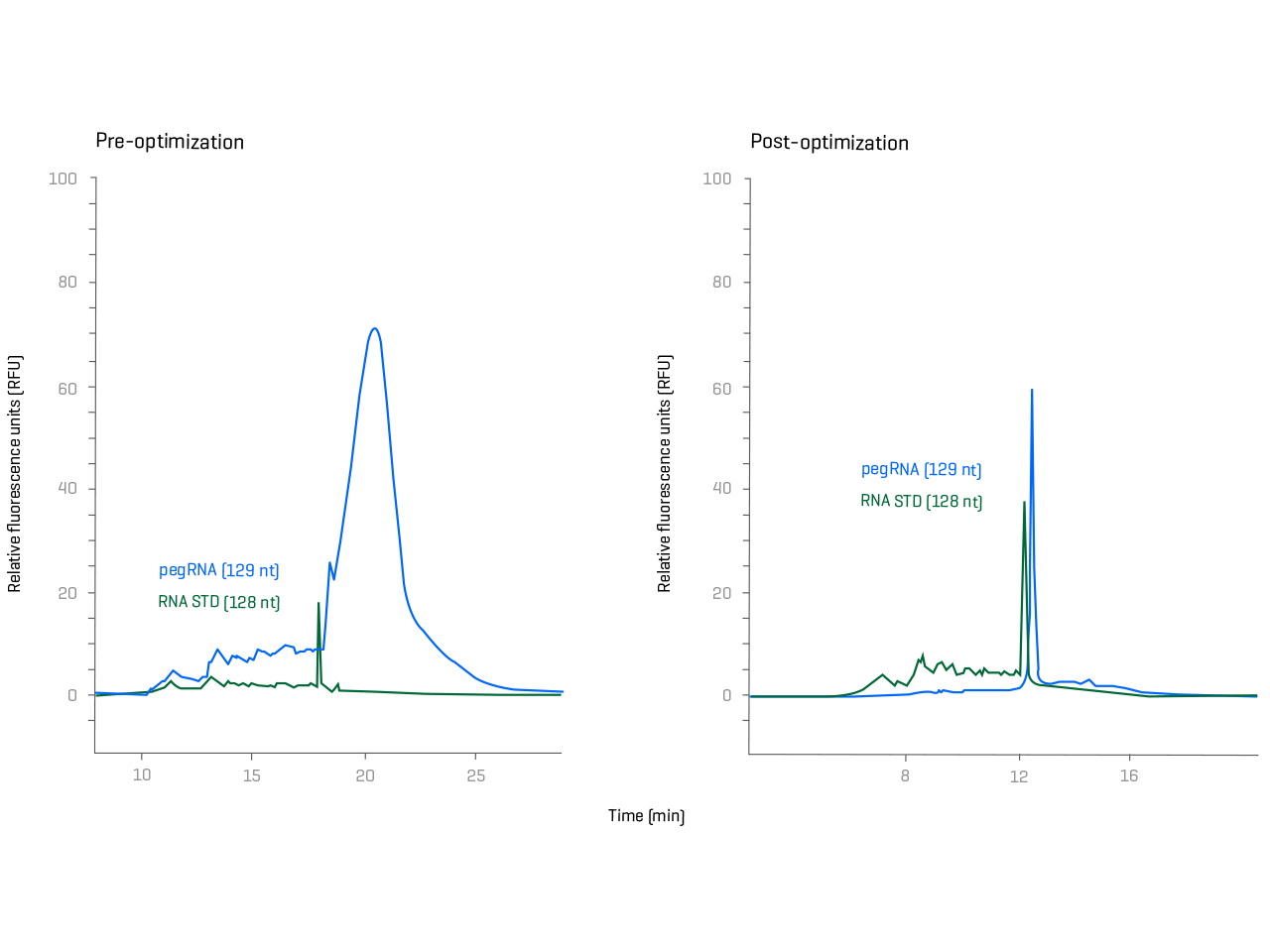
More sophisticated, chemically synthesized gRNAs are in development with the goal of increasing efficiency and specificity of CRISPR/Cas9 gene editing. One of these newly emerging classes is pegRNA with sizes ranging from around 120–250 nucleotides (nt).
The length and associated risk of secondary structure formation, however, pose challenges for the analytical assessment of its purity.
Move beyond these boundaries of CRISPR/Cas9 analysis and confirm size and purity for large pegRNA with confidence. Rely on high resolving power and repeatability to separate your products from impurities with similar properties.
- Reliable purity assessment through high-resolution separation of full-length pegRNAs from their impurities
- Control of secondary structures of chemically synthesized pegRNAs (>100 nt)
- Excellent repeatability with accurate and stable temperature control
- Easy assay transfer to regulated environments due to compatibility with the Empower CDS
Characterize therapeutic molecules with confidence with the kit-based system.
Kit with coated capillaries, gel, and standards to analyze single-stranded nucleic acids on the PA 800 Plus system.
Intact mass determination
Workflow
Synthetic oligonucleotides —such as guide RNAs, sgRNAs and pegRNAs—are prone to a high number of impurities due to their stepwise chemical synthesis. Obtaining molecular weight information to confirm the identity of the main product and quantitation of related impurities is key for quality assessment prior to performing lengthy cell-based assays.
Take control of the purity of your products with high-quality, accurate mass data derived from intuitive solutions. Enable your team to gain key insights that are relevant for ensuring safety and efficacy.
- Excellent spectra quality with superior ionization and declustering of salt adducts using the Turbo V ion source
- A short learning curve with intuitive SCIEX OS software for acquisition and processing
- Additional quantitation and deconvolution options with SCIEX OS software
Intact mass determination
Suited for:
- Straightforward molecular weight determination of sgRNAs and impurities
- Robust, analytical flow setup
Reproducibility, reliability and carryover performance to match your quantitative workflows. Dependability you can count on, from injection to injection and batch after batch.
A purpose-built QTOF system, designed specifically to accelerate everyday biologics characterization.
Unleash the analytical power of the next-generation software platform for data acquisition and processing.
Intact mass determination
Suited for:
- Straightforward molecular weight determination of sgRNAs and impurities
- Detection of low abundance impurities with superior linear dynamic range (LDR)
Reproducibility, reliability and carryover performance to match your quantitative workflows. Dependability you can count on, from injection to injection and batch after batch.
A high-resolution mass spectrometry solution that combines powerful MS/MS sensitivity, fragmentation technology and a step-change in data independent acquisition.
Unleash the analytical power of the next-generation software platform for data acquisition and processing.
Aptamer selection (CE-SELEX)
Workflow
Selection of high-affinity target binders from pools of random aptamer sequences can be a challenging task. Apart from cumbersome processes taking weeks to months, the risk of enriching non-suitable binders and the loss of the strongest binders to surfaces are concerns linked to traditional approaches.
Take back your time with superior workflows to enrich the most promising aptamers for an efficient drug development process.
- Increase the number of viable targets with free solution selection
- Realize optimized aptamer sequence development with reduced sequence motif bias
- Shorten aptamer selection processes from weeks or months to days
- Confidently implement a solution for limited sample amounts during early development
Characterize therapeutic molecules with confidence with the kit-based system.
Pre-assembled bare-fused silica capillary cartridge – 30 cm for the PA 800 Plus system.
Featured resource
-
Technical note
CE-SELEX: Rapid aptamer selection using capillary electrophoresis
Discover how to combine CE with systematic evolution of ligands by exponential enrichment (SELEX) to enable better and more efficient selection of your high-affinity DNA or RNA aptamers.
-
Webinar
CE-SELEX: Capillary electrophoresis-based selection of aptamers
Join professor Michael T. Bowser (Department of Chemistry, University of Minnesota) to learn how to take back your time for aptamer selection, while increasing the number of viable target sequences.
All resources
-
Technical note
Identification and relative quantitation of therapeutic oligonucleotide impurities
Take control of your API and related impurities with intuitive solutions. Discover how to confirm sequences of your product and achieve impurity ID for highly modified nucleic acid drugs.
-
Technical note
Characterization of oligonucleotides and related impurities to support the development of nucleic acid-based therapeutics
Remove barriers to the detection of impurities in your therapeutic oligonucleotide drugs. Explore how to achieve impurity ID with full sequence coverage for highly modified ASOs present at levels as low as 0.3%.
-
Technical note
Molecular weight determination and impurity analysis of a 70-mer oligonucleotide
Imagine taking control of your CRISPR/Cas9 gene-editing systems by ensuring sgRNA integrity and purity. Be empowered to take on the challenge of future medicine development with accurate mass solutions that provide meaningful answers.
-
Webinar
High-resolution size and purity determination of sgRNA and Cas9 mRNA in a single analysis
Learn about an intuitive kit-based solution for the characterization of mRNA integrity and purity. Break through boundaries with workflows that provide the highest resolution for sgRNA and mRNA in a single analysis.
-
Technical note
Compliant-ready, streamlined data management for Cas9 mRNA analyses with Empower Chromatography Data System
Set your own schedule for mRNA analyses by streamlining your CE data management with the Empower CDS.
-
Webinar
Empowering next-generation therapy development with streamlined data integration
Discover how to save time by streamlining your CE data management with the Empower CDS.
-
Technical note
Comprehensive method development for a wide size range of single-stranded nucleic acids
Create pathways for effective method development and learn how to achieve the highest quality data for RNA products with sizes ranging from 50–9,000 nucleotides and beyond.
-
Technical note
Analysis of pegRNA
Overcome secondary structure challenges presented by large-sized oligonucleotides for gene editing. Learn how to enable accurate and easy purity analysis of complex synthetic pegRNAs.
-
Technical note
CE-SELEX: Rapid aptamer selection using capillary electrophoresis
Discover how to combine CE with systematic evolution of ligands by exponential enrichment (SELEX) to enable better and more efficient selection of your high-affinity DNA or RNA aptamers.
-
Webinar
CE-SELEX: Capillary electrophoresis-based selection of aptamers
Join professor Michael T. Bowser (Department of Chemistry, University of Minnesota) to learn how to take back your time for aptamer selection, while increasing the number of viable target sequences.
-
Technical note
Visualize intact oligonucleotide data for rapid sample quality and impurity assessment
Discover a user friendly workflow for rapid and confident identification, impurity assessment, and characterization of synthetic oligonucleotides, with software that provides flexible tools for sequence creation, deconvolution, and mass mapping.
Associated applications
Advance the development of your next therapeutic drug with easy-to-operate, integrated analytical systems that can provide new insights into ADME-Tox and DMPK. Rapidly and comprehensively characterize drug candidates with robust and sensitive solutions for identification, monitoring and quantitation.
Set your own schedule for CRISPR/Cas9 gene editing with innovative, intuitive analytical solutions. Unleash the potential of your Cas9 messenger RNA (mRNA), single guide RNA (sgRNA) and Cas9 proteins by understanding quality, purity and safety and assessing on/off target effects.
Expand the possibilities of drug delivery with innovative analytical solutions for lipid raw materials, LNPs and other non-viral carriers. Move beyond boundaries in structural elucidation and monitoring of analytes to estabilish confidence in the quality and stablity of your products.
Advance your drug discoveries with reliable metabolite data that save time and money. Our range of solutions supports routine monitoring and complex identification to give your team confidence during synthetic therapeutic development.